Healthcare providers continue to present patients with more personalized risk assessments, diagnoses, and treatments. Due to advances in precision medicine, clinicians are increasingly moving away from a traditional “one size fits all” perspective and towards individualized healthcare decisions for their patients.
Precision medicine is an approach to healthcare that uses personal information—including genetic, environmental, and lifestyle data—to improve the prevention, diagnosis, and treatment of disease.[1]
From diagnosis to treatment, tools such as imaging data, artificial intelligence (AI), advanced clinical applications, biomarkers, and therapeutics are especially poised to help inform clinicians’ personalized approach to each patient’s care.
Advancing precision medicine across the patient care journey
Precision imaging can assist in diagnosing disease with enhanced accuracy, helping clinicians better delineate and identify prognoses, and help inform treatments for oncological and non-oncological disorders. Precision imaging is descriptive, predictive, and integrative—effectively moving acquired data to clinical insights.[2]
Medical image analysis has grown into a mature field by continually challenging limits across imaging technologies such as computed tomography (CT) and magnetic resonance imaging (MRI), along with more recent breakthroughs in molecular imaging.[3] Imaging data is a critical component of the clinical information that can enable precision diagnostics and therapeutics, leading to newer techniques such as theranostics.
With more precise and integrated care, imaging diagnostics and therapeutics are impacting clinical outcomes across the patient care journey, in areas such as prostate cancer, breast cancer, and more.
Achieving precision medicine with AI and precision diagnostics across a shifting patient population
The number of patients in need of specialized care is growing. According to the World Health Organization, the number of people 80 years and older will triple between 2020 and 2050.[4] The current population also has a large number of chronic diseases—for example, in the U.S., six in ten adults experience chronic disease, and four in ten have two or more chronic diseases.[5]
The convergence of AI and precision medicine is promising to revolutionize healthcare for this evolving patient population, enabling those with less‐common responses to treatment or unique healthcare needs to get the right care at the right time. This can be achieved when robust AI algorithms generate insights and empower clinician decision-making through augmented intelligence.[6] Leaders such as GE Healthcare are committed to advancing precision medicine by integrating clinical care and data across the patient journey.
“We’re focused on supporting clinicians as they sustain the health and well-being of their patients; we’re continuing to innovate medical imaging and precision diagnostics to help improve clinical outcomes and increase access to care,” said Jean-Luc Procaccini, President & CEO, Molecular Imaging & Computed Tomography, GE Healthcare.
“As AI is introduced to aggregate important health data—such as data from preventative screenings, advanced precision diagnostics, and therapeutics efforts—it will enable further insights to inform clinical decisions for diagnosis and treatment.”
Employing precision imaging technology for better clinical insights
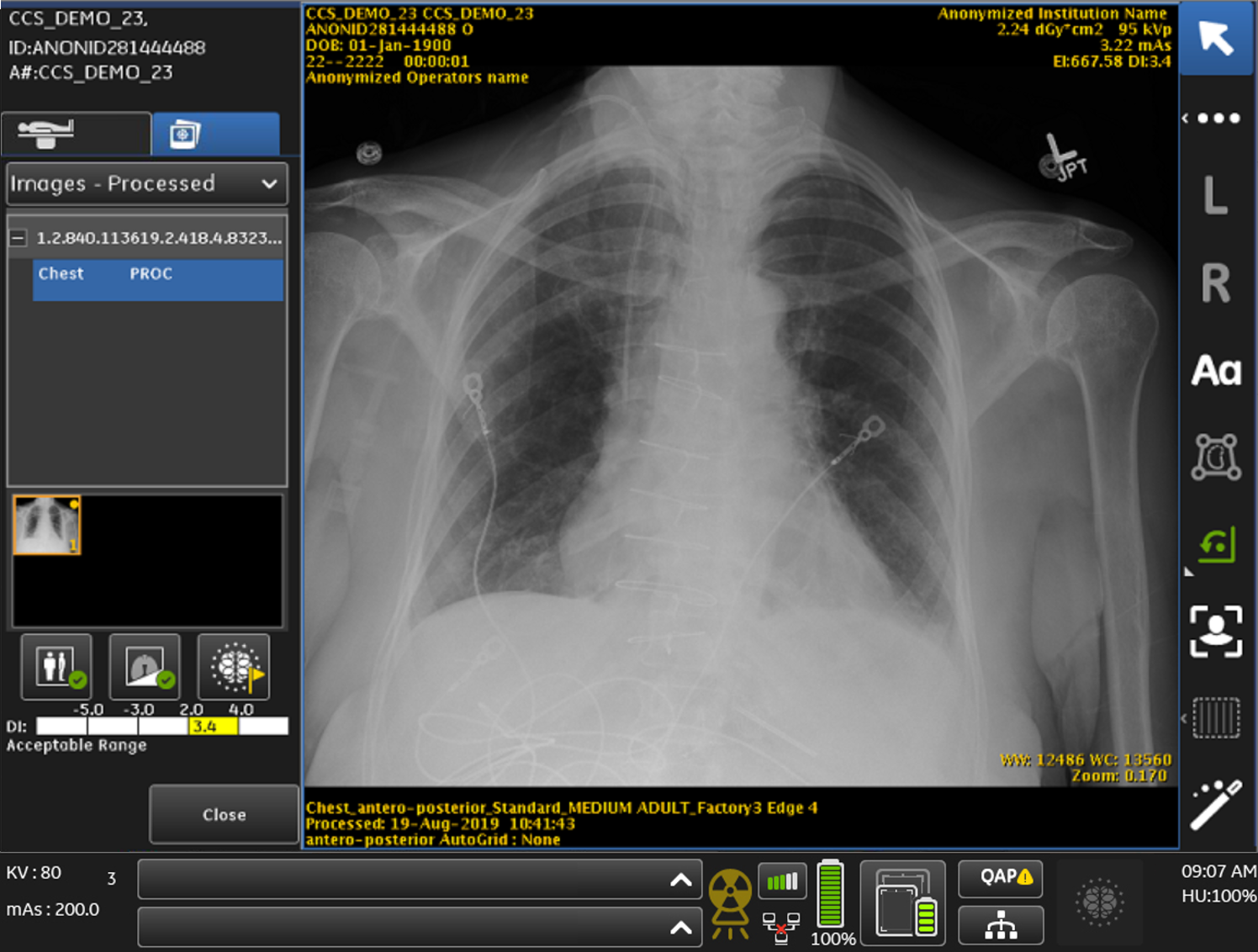
As powerful diagnostic tools, medical imaging systems offer clinicians an information-rich opportunity to image and evaluate disease in clinical areas such as oncology and neurology. The widespread application of AI across imaging modalities such as X-ray, MRI, and molecular imaging supports efforts to improve diagnostics, treatment, and follow-up care.
Incorporating AI technologies in medical imaging helps radiologists process large volumes of imaging data with advanced image reconstruction algorithms and can help improve the consistency and accuracy of imaging data by providing clearer and sharper images that can support a more precise diagnosis for patients. By using AI technologies to analyze images gathered during a scan, clinicians may be able to identify conditions more quickly, promoting early intervention.
For example, a robust collection of on-device X-ray AI algorithms help automate measurements, prioritize cases, and ensure quality control. It can assist technologists with correct anatomical patient positioning and protocol selections at the time of the scan for improved data consistency. AI can also analyze images upon acquisition for critical findings, like pneumothorax and flag the most urgent cases for prioritized radiologist review.
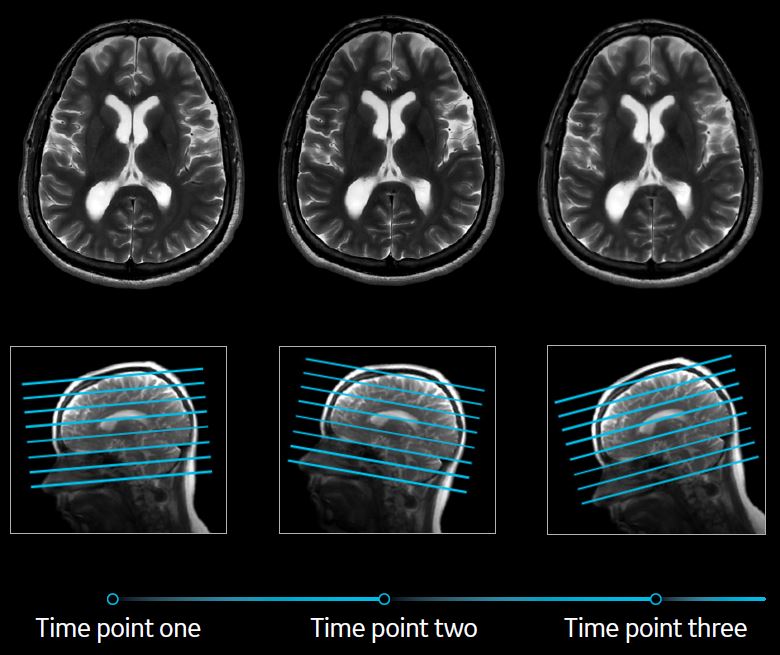
Multiple scans are often needed to monitor disease over time. For neurological MRI studies, specifically, to be evaluated longitudinally, they must image the same area of interest each time. The processes of result validation and reproducibility of different neuroimaging analyses and statistical maps in MRI are often complex due to several factors—some of which include changes in the patient between scans, presence of noise in the imaging data, and variations in study designs, sample sizes, and sampling protocols.[7]
An automated MR workflow tool that uses state-of-the-art AI to precisely identify and align MRI scans for diagnostic neuroimaging is now available. It automates the area of interest and enables consistent, repeatable scan alignment to help physicians monitor a patient across longitudinal studies that may be several months apart. It can also help reduce the set-up time for MRI studies and helps produce images with less variability between technologists and scans, reducing the chances that a patient will be recalled due to incorrect slice placement.
Advancing the integration of diagnostics and precision therapies with theranostics
In addition to powerful innovations in imaging technology, molecular imaging has rapidly improved diagnostic capabilities as well due to the discovery and development of new tracers. New imaging tracers and targeted therapeutic pairings—working with sophisticated detection technology—make advanced diagnoses and targeted treatments possible, thereby reinforcing molecular medicine’s key role in precision diagnostics and therapeutics.
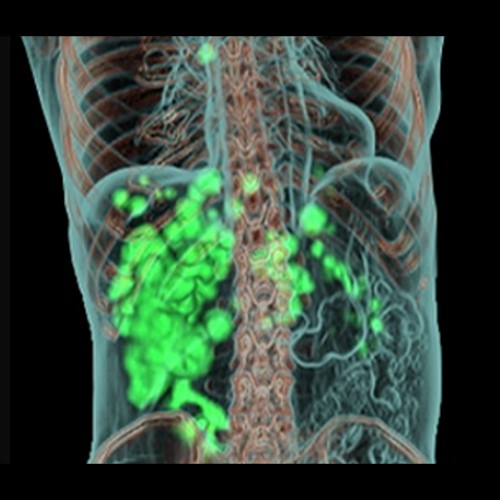
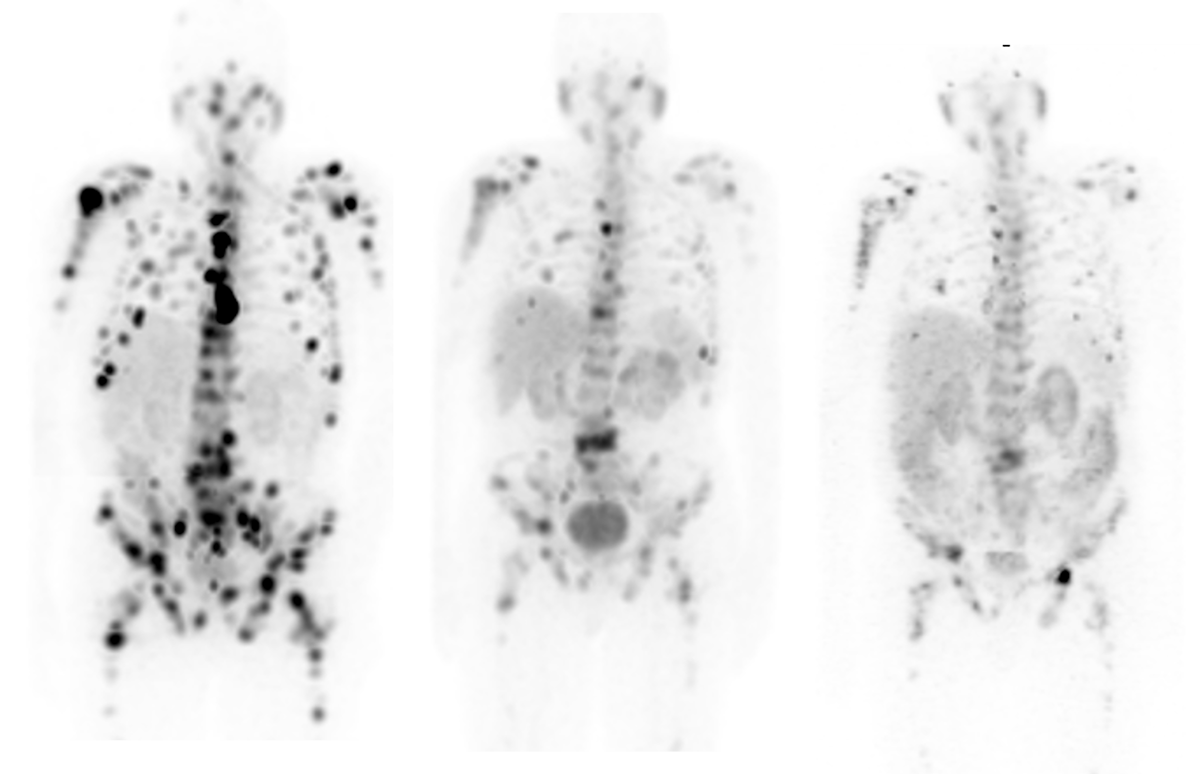
Theranostics is an exciting field of molecular medicine that leverages the tumor detection properties of conventional diagnostic radiotracers to deliver radiotherapy. Enabled by molecular imaging techniques such as positron emission tomography (PET) and single photon emission tomography (SPECT), the theranostic approach uses specific targeting compounds for both imaging and therapy of a particular malignancy. Its ability to identify areas of disease that are more likely to respond to targeted therapies is invaluable to cancer patients. Moreover, clinicians are praising the theranostic approach’s potential to help with more effective disease management by aligning patients with treatments that will be most impactful for them. Additional innovations in theranostics have the potential to:
- Introduce much more imaging data for processing.
- Include highly sophisticated automated tools and AI-based reconstruction algorithms.
- Involve digital solutions that allow for dosimetry on theranostic patients.
- Increase the use of imaging to assess treatment efficacy.
The market size for theranostics applications in oncology is set to expand from $73.1B in 2020 to $119.B by 2025 and is considered one of the best new approaches for early identification, treatment, and monitoring of metastatic, heterogeneous, and recurrent diseases.[8] Molecular imaging is essential in theranostics, allowing for non-invasive, repetitive assessment of the compound uptake and characterization of the tumor tissue and therapy response over time.
New theranostic target pairs have been developed, validated, and successfully used in treating lymphomas, neuroblastoma, neuroendocrine tumors, and, more recently, certain prostate cancers. Strong clinical needs in areas such as prostate and other cancers continue to fuel the search for additional diagnostic and therapeutic pairings with the goal of improving the quality of life and outcomes for cancer patients. These opportunities enable researchers and clinicians to bring more targeted tracers to the market and the patient.
Committed to a future of precision medicine led by precision imaging
Building a stronger future for precision health requires staying ahead of the pace at which the environment continues to change and continually challenging the traditional clinical and operational models. More precise diagnostic tools, consistent imaging data, and robust AI tools are needed to improve patient care and outcomes and develop meaningful insights derived from probing the terabytes of data generated every day.
GE Healthcare has been delivering robust solutions that support clinicians in medical imaging—advancing both imaging technology as well as the supporting systems and solutions that enable improved healthcare. GE Healthcare will continue its commitment to building on these innovations with state-of-the-art technology and AI-based solutions to advance precision medicine.
Learn more about GE Healthcare’s precision diagnostic and therapeutic imaging offerings.
DISCLAIMER
Not all products or features are available in all geographies. Check with your local GE Healthcare representative for availability in your country.
REFERENCES
[1] https://www.brookings.edu/research/advancing-precision-medicine-through-agile-governance/#:~:text=Precision%20medicine%20is%20an%20approach,diagnosis%2C%20and%20treatment%20of%20disease.
[2] https://www.sciencedirect.com/science/article/pii/S1361841516301049
[3] Frangi AF, Taylor ZA, Gooya A. Precision Imaging: more descriptive, predictive and integrative imaging. Med Image Anal. 2016 Oct;33:27-32. doi: 10.1016/j.media.2016.06.024. Epub 2016 Jun 23. PMID: 27373145.
[4] https://www.who.int/news-room/fact-sheets/detail/ageing-and-health
[5] https://bmcpublichealth.biomedcentral.com/articles/10.1186/s12889-019-7762-5#:~:text=The%20ageing%20population%20tends%20to,of%2056.3%25%20%5B11%5D.
[6] Johnson KB, Wei WQ, Weeraratne D, Frisse ME, Misulis K, Rhee K, Zhao J, Snowdon JL. Precision Medicine, AI, and the Future of Personalized Health Care. Clin Transl Sci. 2021 Jan;14(1):86-93. doi: 10.1111/cts.12884. Epub 2020 Oct 12. PMID: 32961010; PMCID: PMC7877825.
[7] Dinov ID. Neurological imaging: statistics behind the pictures. Imaging Med. 2011;3(4):423-432. doi:10.2217/iim.11.37
[8] https://www.globenewswire.com/news-release/2020/10/26/2114163/0/en/Global-Theranostics-Markets-Report-2020-Oncology-Segment-Market-for-Theranostics-Should-Grow-from-73-1-Billion-in-2020-to-119-7-Billion-by-2025.html