INDICATIONS AND USAGE:
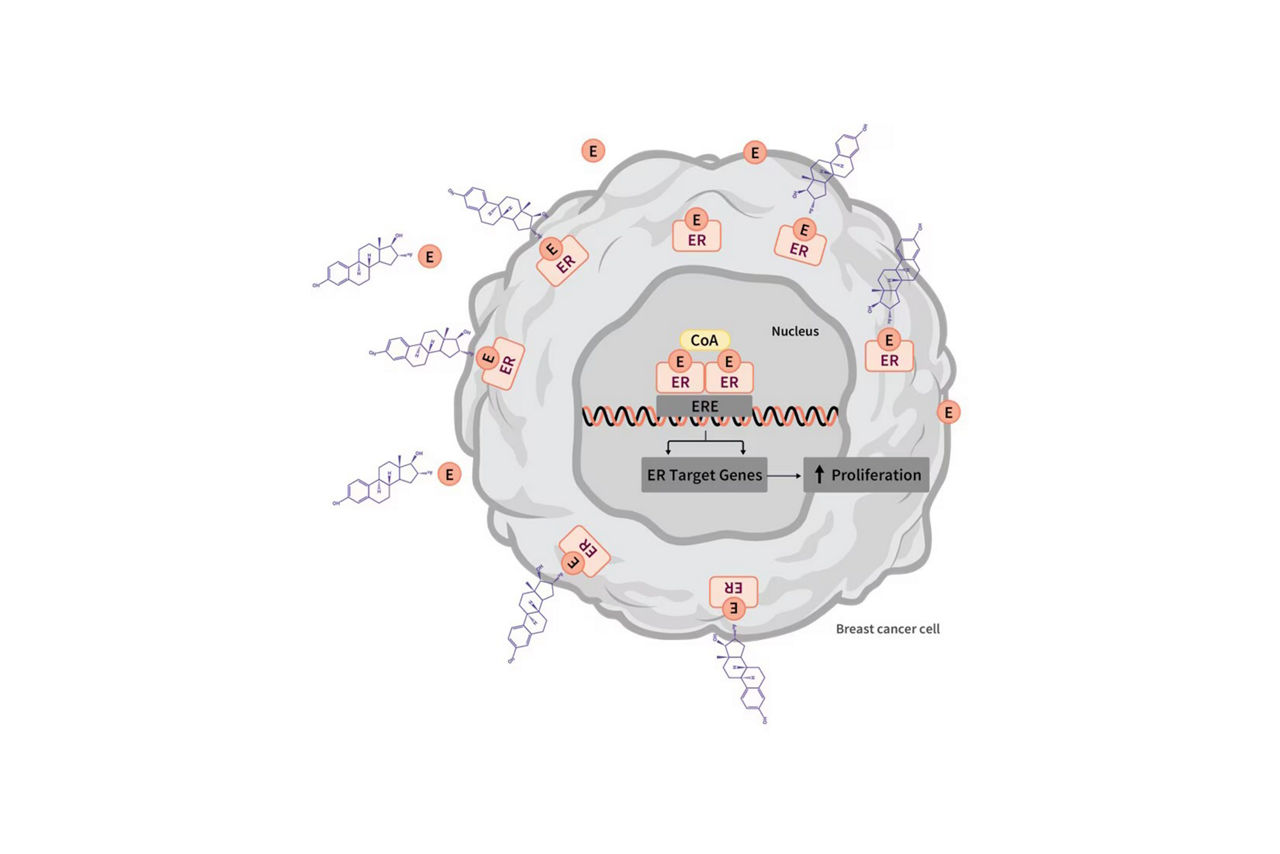
CERIANNA is indicated for use with positron emission tomography (PET) imaging for the detection of estrogen receptor (ER)-positive lesions as an adjunct to biopsy in patients with recurrent or metastatic breast cancer.
Limitations of Use:
Tissue biopsy should be used to confirm recurrence of breast cancer and to verify ER status by pathology. CERIANNA is not useful for imaging other receptors, such as human epidermal growth factor receptor 2 (HER2) and the progesterone receptor (PR).
Important Safety Information
CONTRAINDICATIONS
WARNINGS AND PRECAUTIONS
Risk of Misdiagnosis
Inadequate Tumor Characterization and Other ER-Positive Pathology
- Breast cancer may be heterogeneous within patients and across time. CERIANNA images ER and is not useful for imaging other receptors such as HER2 and PR. The uptake of fluoroestradiol F 18 is not specific for breast cancer and may occur in a variety of ER-positive tumors that arise outside of the breast, including from the uterus and ovaries. Do not use CERIANNA in lieu of biopsy when biopsy is indicated in patients with recurrent or metastatic breast cancer.
False Negative CERIANNA Scan
- A negative CERIANNA scan does not rule out ER-positive breast cancer. Pathology or clinical characteristics that suggest a patient may benefit from systemic hormone therapy should take precedence over a discordant negative CERIANNA scan.
Radiation Risks
- Diagnostic radiopharmaceuticals, including CERIANNA, expose patients to radiation. Radiation exposure is associated with a dose-dependent increased risk of cancer. Ensure safe drug handling and patient preparation procedures (including adequate hydration and voiding) to protect patients and health care providers from unintentional radiation exposure.
Pregnancy Status
- Assessment of pregnancy status is recommended in females of reproductive potential before administering CERIANNA.
ADVERSE REACTIONS
- In Clinical Trials (n=1207) the most common adverse reactions seen occurred at a rate < 1% were injection-site pain and dysgeusia.
USE IN SPECIFIC POPULATIONS
Pregnancy:
Risk Summary
- All radiopharmaceuticals, including CERIANNA, have the potential to cause fetal harm depending on the fetal stage of development and the magnitude of radiation dose. Advise a pregnant woman of the potential risks of fetal exposure to radiation from administration of CERIANNA.
- There are no available data on CERIANNA use in pregnant women. No animal reproduction studies using fluoroestradiol F 18 have been conducted to evaluate its effect on female reproduction and embryo-fetal development.
- The estimated background risk of major birth defects and miscarriage for the indicated populations is unknown. All pregnancies have a background risk of birth defects, loss, or other adverse outcomes. In the U.S. general population, the estimated background risk of major birth defects and miscarriage in clinically recognized pregnancies is 2-4% and 15-20%, respectively.
Lactation:
Risk Summary
- There are no data on the presence of fluoroestradiol F 18 in human milk, or its effects on the breastfed infant or milk production. Lactation studies have not been conducted in animals. Advise a lactating woman to avoid breastfeeding for 4 hours after CERIANNA administration in order to minimize radiation exposure to a breastfed infant.
Pediatric Use
- The safety and effectiveness of CERIANNA in pediatric patients have not been established.
Geriatric Use
- Clinical studies of fluoroestradiol F 18 injection did not reveal any difference in pharmacokinetics or biodistribution in patients aged 65 and over.
DRUG INTERACTIONS
Systemic Endocrine Therapies that Bind to ER
- Drugs that bind to the ER, including SERMs and SERDs, may compete with the binding of fluoroestradiol F18 and may reduce detection of ER-positive lesions with CERIANNA.
- Before administration of CERIANNA, discontinue drugs that bind to the ER, such as SERMs and SERDs, for at least 5 biological half-lives: ( e.g. elacestrant for 11 days, tamoxifen for 8 weeks and fulvestrant for 28 weeks).
To report SUSPECTED ADVERSE REACTIONS, contact GE HealthCare at 800 654 0118 (option 2 then option 1) or by email at GPV.drugsafety@gehealthcare.com or FDA at 800 FDA 1088 or www.fda.gov/medwatch