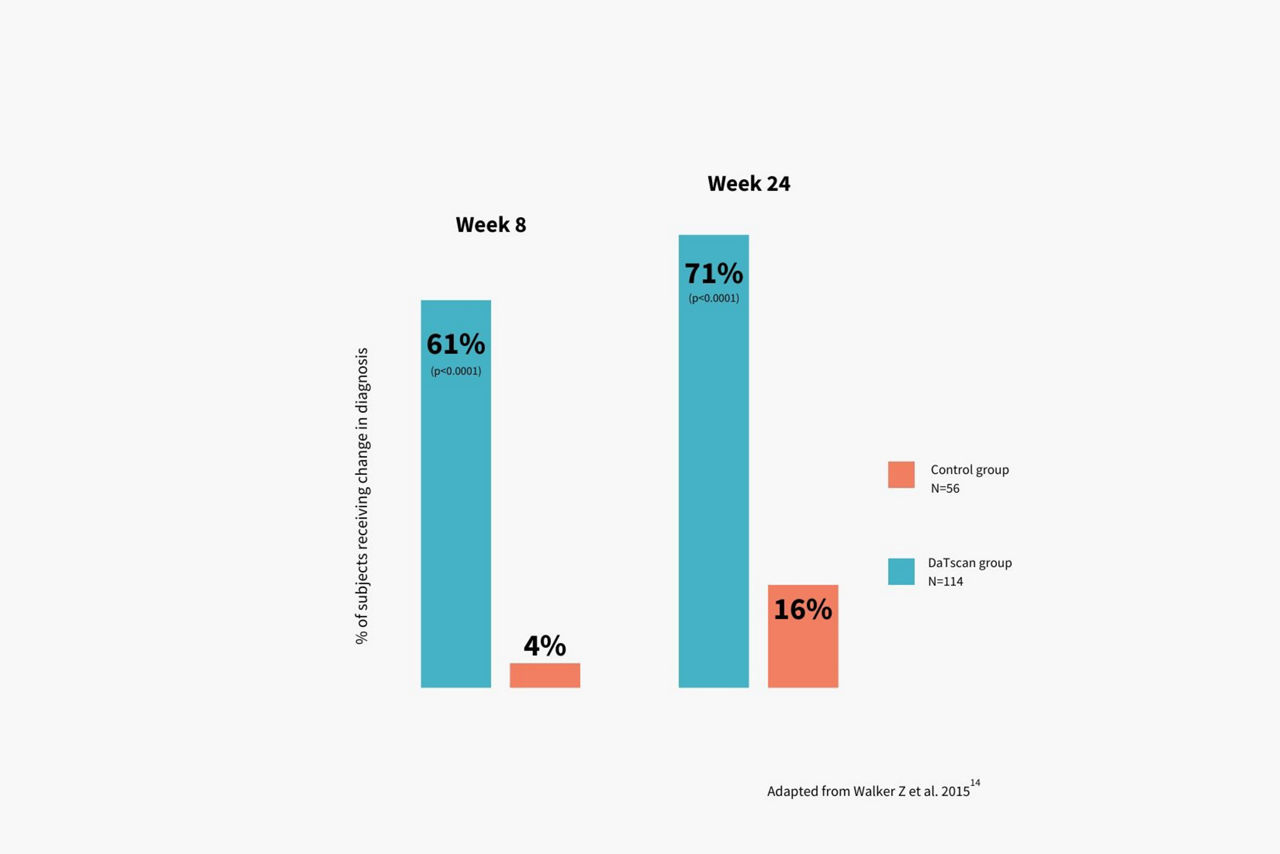
Product Indications and Important Safety Information – DaTscan
PRODUCT INDICATION AND USAGE
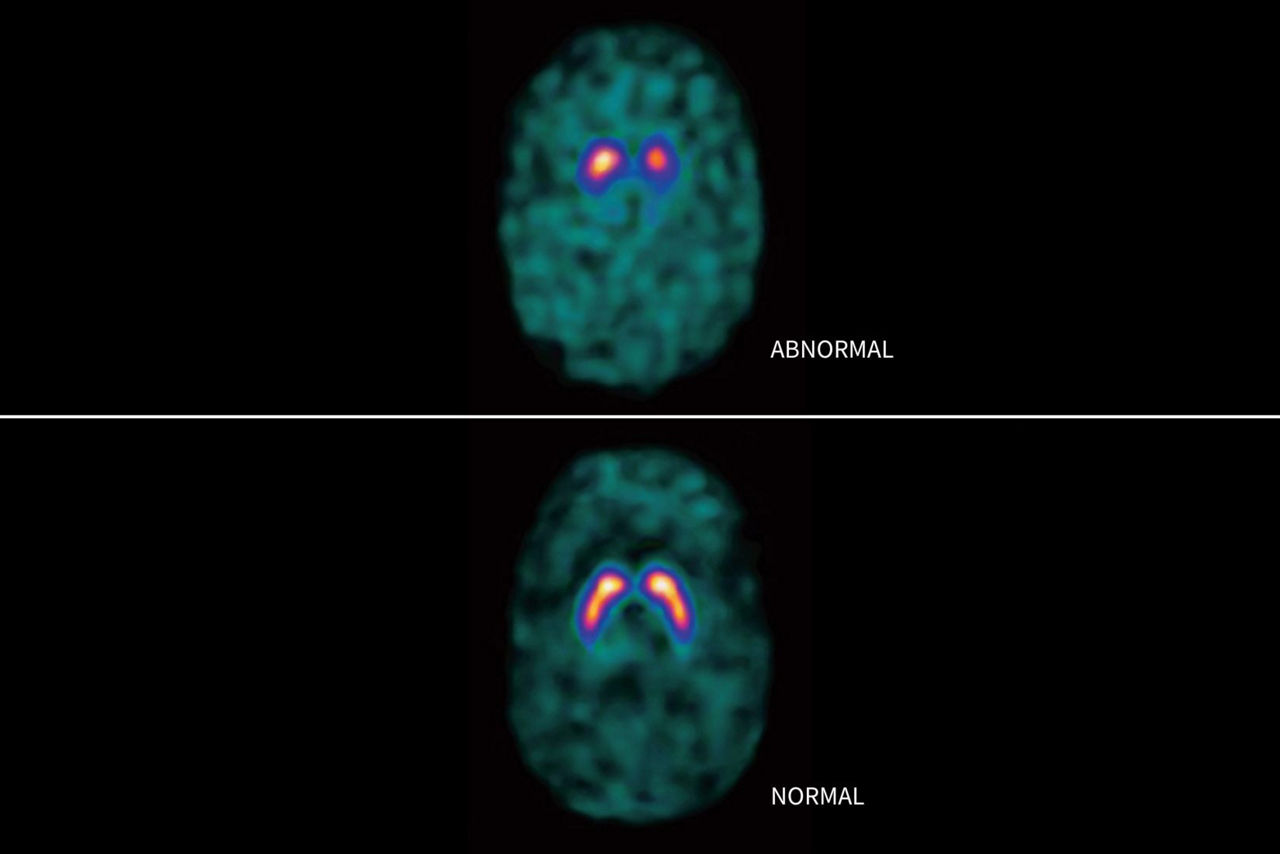
DATSCAN is indicated as an adjunct to other diagnostic evaluations for striatal dopamine transporter visualization using single photon emission computed tomography (SPECT) brain imaging in adult patients with:
• suspected Parkinsonian syndromes (PS) or
• suspected dementia with Lewy bodies (DLB).
CONTRAINDICATIONS
• DaTscan is contraindicated in patients with known serious hypersensitivity to ioflupane I 123.
WARNINGS AND PRECAUTIONS
• Hypersensitivity Reactions: Hypersensitivity reactions, including dyspnea, edema, rash, erythema, and pruritus, have been reported following DaTscan administration.
• Thyroid Accumulation of Iodine-123: DaTscan may contain up to 6% of free iodide (iodine-123). Thyroid uptake of iodine-123 may result in an increased long-term risk for thyroid neoplasia. To decrease thyroid accumulation of iodine-123, block the thyroid gland before administration of DaTscan.
• Radiation Risk: DaTscan contributes to patient’s overall long-term cumulative radiation exposure. Long-term cumulative radiation exposure is associated with an increased risk for cancer. Ensure safe handling to minimize radiation exposure to patients and healthcare providers.
ADVERSE REACTIONS
• In clinical trials, headache, nausea, vertigo, dry mouth, or dizziness were reported at a rate of 1% or less. In postmarketing experience, hypersensitivity reactions and injection-site pain have been reported.
DRUG INTERACTIONS
• Drugs that bind to the dopamine transporter with high affinity have the potential to impact DaTscan image interpretation. The impact of dopamine agonists and antagonists upon DaTscan imaging results has not been established.
• In patients taking venlafaxine or desvenlafaxine prior to DaTscan administration, there have been cases of altered DaTscan uptake that impacted image interpretation. The mechanism of interference of these drugs with DaTscan imaging is not known.
USE IN SPECIFIC POPULATIONS
• Pregnancy: Radioactive iodine products cross the placenta and can permanently impair fetal thyroid function. Administration of a thyroid blocking agent is recommended before the use of DaTscan in a pregnant woman. All radiopharmaceuticals have potential to cause fetal harm. There are no available data on DaTscan use in pregnant women to evaluate for a drug-associated risk of major birth defects, miscarriage or adverse maternal or fetal outcomes. Advise pregnant woman of the potential risks of fetal exposure to radiation with the administration of DaTscan.
• Lactation: Iodine 123 (I 123), the radionuclide in DaTscan, is present in human milk. There is no information on the effects on breastfed infants or on milk production. Advise a lactating woman to interrupt breastfeeding and pump and discard breast milk for at least 6 days after DaTscan administration to minimize radiation exposure to a breastfeeding infant.
• Pediatric Use: The safety and efficacy of DaTscan have not been established in pediatric patients.
• Geriatric Use: There were no overall differences in safety and effectiveness between elderly patients, aged 65 and older, and younger patients.
• Renal Impairment: DaTscan is excreted by the kidney and patients with severe renal impairment may have increased radiation exposure and altered DaTscan images.
OVERDOSAGE
• The risks of overdose relate predominantly to increased radiation exposure, with the long-term risks for neoplasia. In case of overdosage of radioactivity, frequent urination and defecation should be encouraged to minimize radiation exposure to the patient.
Prior to DaTscan administration, please read the full Prescribing Information for additional Important Safety Information.
To report SUSPECTED ADVERSE REACTIONS, contact GE HealthCare at 800 654 0118 (option 2, then option 1) or the FDA at 800 FDA 1088 or www.fda.gov/medwatch