Don't have an account?
At a glance
Safety Simplicity Image Quality in every vial.
Your UEA of Choice
Diagnostic confidence
Impact of Optison on the assessment of regional wall motion (N=176 patients)10
The FDA has approved Optison for pediatric patients with suboptimal echocardiograms.
Optison remains the only PEG-free ultrasound-enhancing agent cleared in the United States for both adults and children, backed by a well-established5,7 record of safety and efficacy.
Because when clarity matters most, Optison is On Your Side.
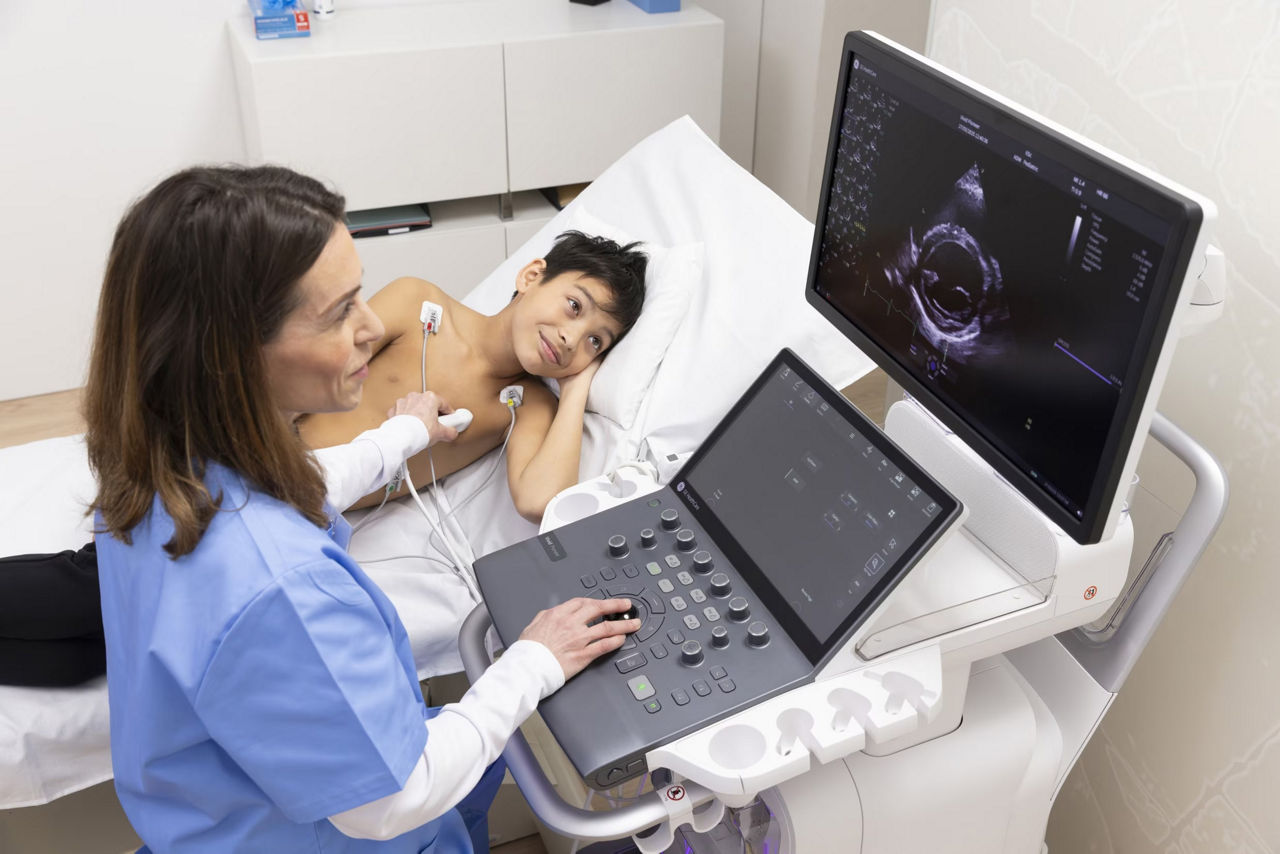
For more than two decades, Optison has supported care teams across diverse patient populations—because when it comes to diagnostic excellence, every detail matters. That’s why Optison offers distinct advantages designed to protect patients and simplify workflows, from avoiding certain hypersensitivity risks to eliminating extra screening steps.


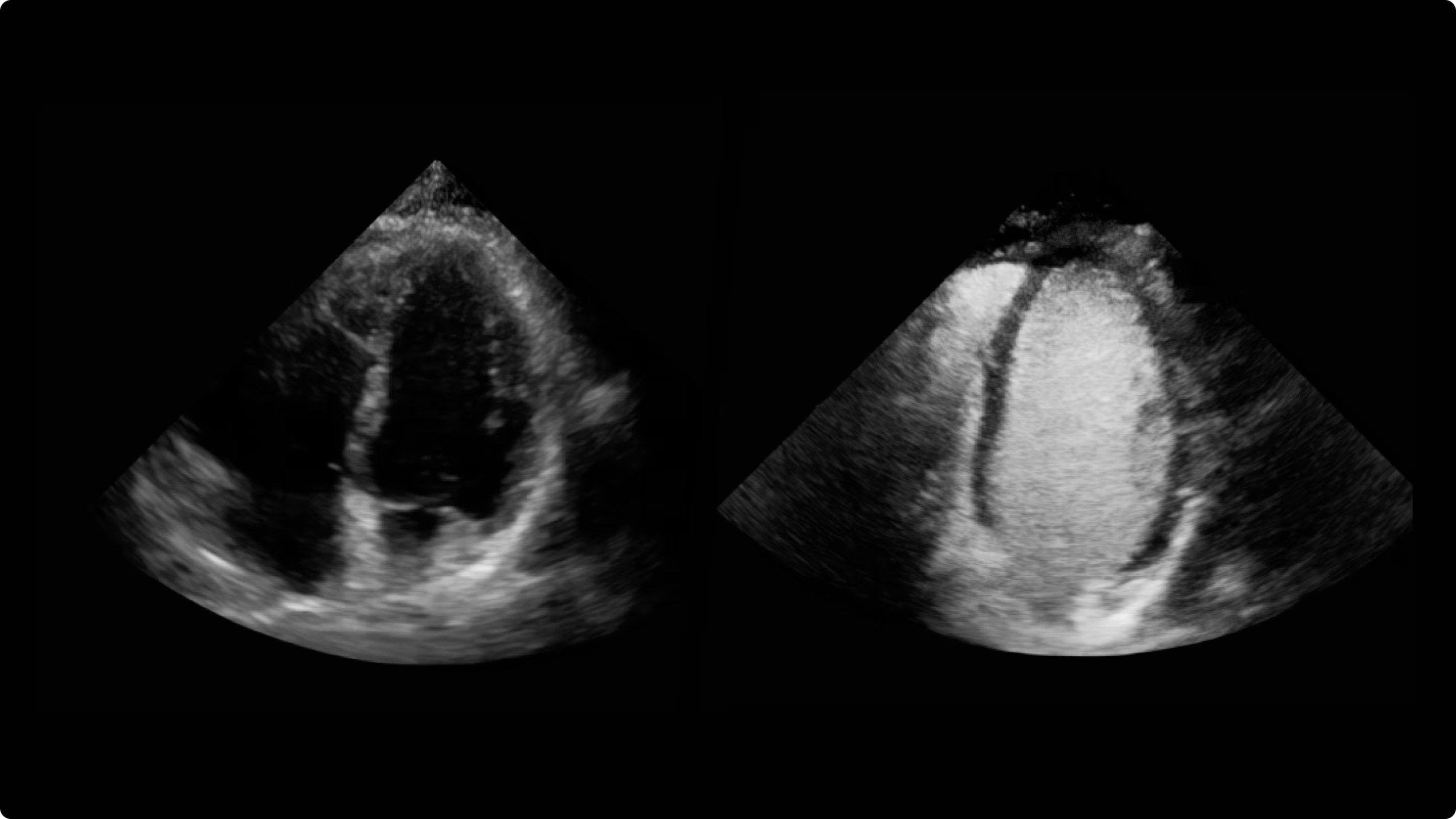
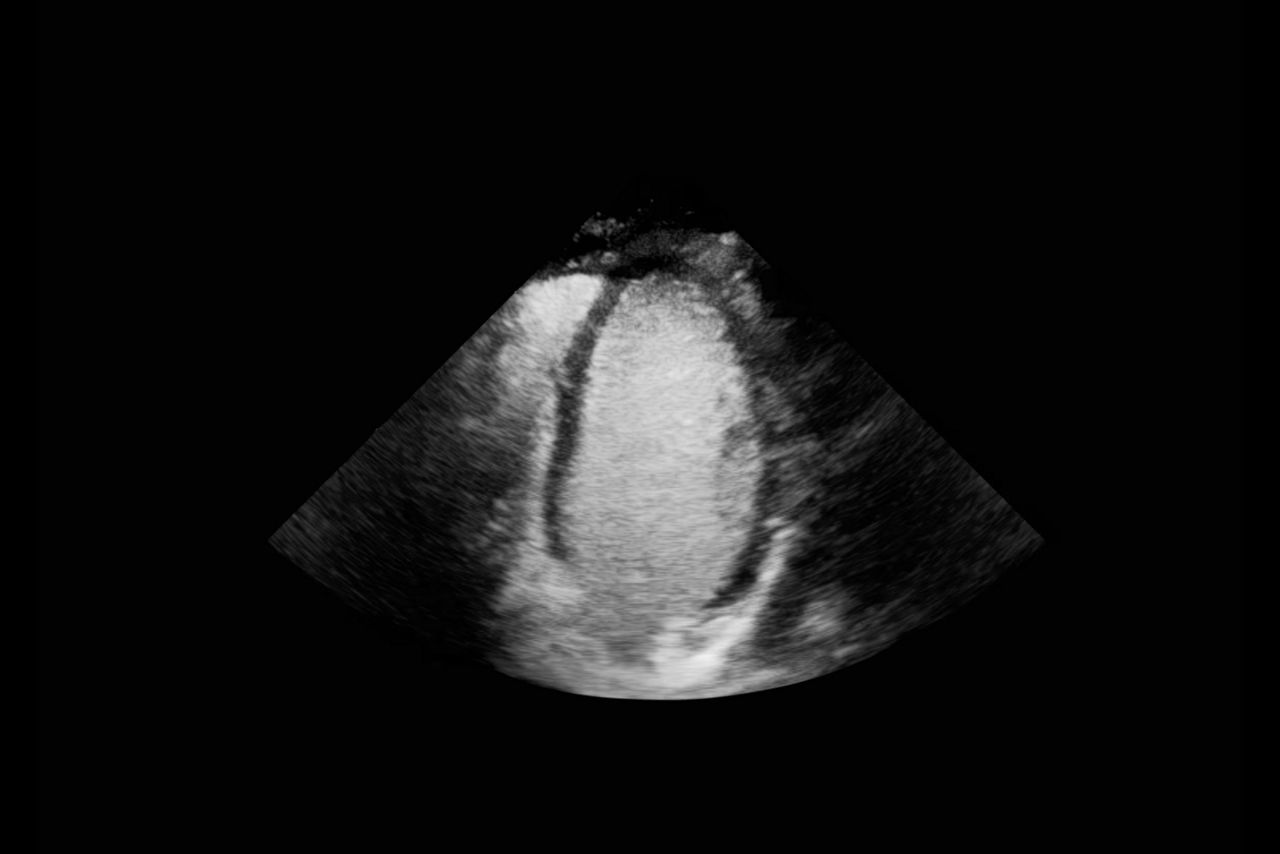
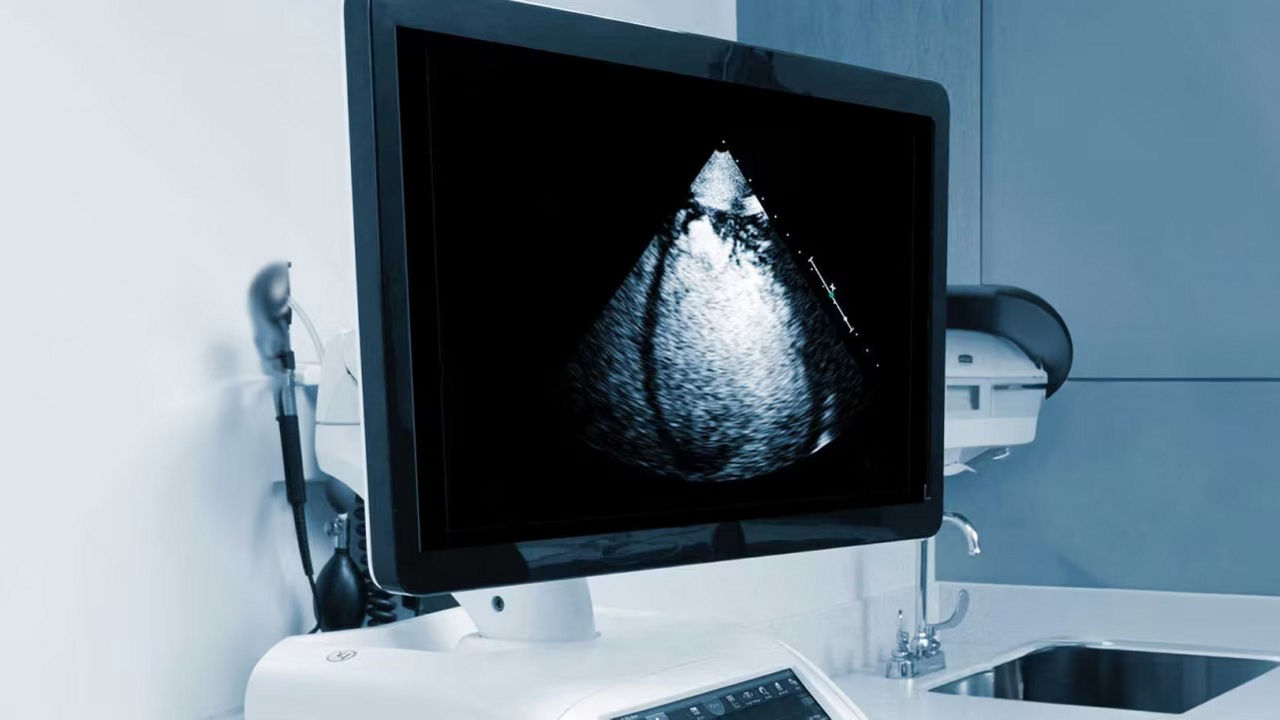
Clear delineation of the endocardial borders may help facilitate evaluation of LV wall motion and function.
a) Images obtained with Vivid™ S70 scanner using Optison.
b) While the products can be used together, Optison and Vivid™ S70 are not Combination Products per 21 CFR 3.2(e); each was FDA-approved independently and neither requires the other.
Optison can integrate seamlessly into your workflow, with less than 60 seconds from suspension to injection⁵
Structure
Optison: The albumin- shelled microsphere
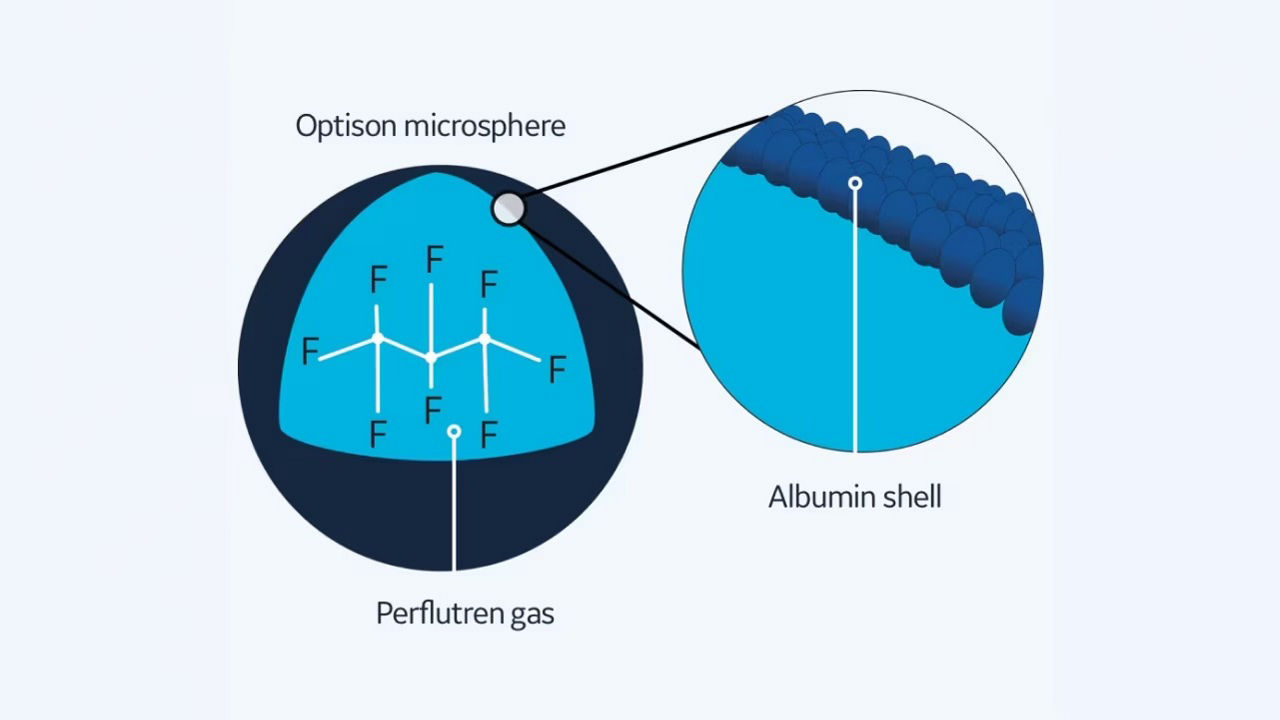
An abundant plasma protein manufactured in the liver
References
- Exuzides A, Main ML, Colby C, Grayburn PA, Feinstein SB, Goldman JH. A retrospective comparison of mortality in critically ill hospitalized patients undergoing echocardiography with and without an ultrasound contrast agent. J Am Coll Cardiol Img. 2010;3:578-585.
- Levitt DG and Levitt MD. Int J Gen Med. 2016;9:229-255.
- Main M, Grayburn P, Lang R, et al. Effect of Optison on pulmonary artery systolic pressure and pulmonary vascular resistance. Am J Cardiol. 2013;112(10):1657-1661.
- Mulvagh SL, Rakowski H, Vannan MA, et al. American Society of Echocardiography Consensus Statement on the Clinical Applications of Ultrasonic Contrast Agents in Echocardiography. J Am Soc Echocardiogr. 2008;21:1179-1201.
- Optison Prescribing Information. Arlington Heights, IL: GE HealthCare, 2025.
- Reilly JP, Tunick PA, Timmermans RJ, et al. Contrast Echocardiography Clarifies Uninterpretable Wall Motion in intensive Care Unit Patients. J Am Coll Cardiol. 2000;35(2):485-490.
- Sabati A, Wang S, Shivaram P, et al. A Phase 4, Open-Label, Non Randomized, Multicenter Study to Evaluate Efficacy (and Safety) of Intravenous Administration of Optison for Contrast-Enhanced Echocardiography in Pediatric Patients. Pediatric Cardiol. (2025). https://doi.org/10.1007/s00246-025-03993-4
- Senior R, Dwivedi G, Hayat S, Keng Lim T. Clinical benefits of contrast-enhanced echocardiography during rest and stress examinations. Eur J Echocardiogr. 2005;6(Suppl.2):S6-S13.
- Wu M, Fields JJ, Sachdev V, et al. Increased susceptibility for adverse reactions to ultrasound enhancing agents in sickle cell disease. J Am Soc Echocardiogr. 2023;36:208-15.
- Zhao H, O’Quinn R, Ambrose, et al. Contrast-Enhanced Echocardiography Has the Greatest Impact in Patients With Reduced Ejection Fractions. J Am Soc Echocardiogr. 2018;31(3):289-296.
Serious cardiopulmonary reactions, including fatalities, have occurred uncommonly during or following perflutren-containing microsphere administration. Most serious reactions occur within 30 minutes of administration
• Assess all patients for the presence of any condition that precludes OPTISON administration
• Always have resuscitation equipment and trained personnel readily available
OPTISON (Perflutren Protein-Type A Microspheres Injectable Suspension, USP)
INDICATIONS:
OPTISON is an ultrasound contrast agent indicated for use in adult and pediatric patients with suboptimal echocardiogram to opacify to the left ventricle to improve the delineation of the left ventricle endocardial borders.
CONTRAINDICATIONS:
OPTISON is contraindicated in patients with known or suspected hypersensitivity to perflutren or albumin.
WARNINGS AND PRECAUTIONS:
- Serious cardiopulmonary reactions including fatalities have occurred uncommonly during or shortly following perflutren-containing microsphere administration, typically within 30 minutes of administration. The risk for these reactions may be increased among patients with unstable cardiopulmonary conditions (acute myocardial infarction, acute coronary artery syndromes, worsening or unstable congestive heart failure, or serious ventricular arrhythmias).
- Serious anaphylactic reactions have been observed during or shortly following perflutrencontaining microsphere administration, including shock, hypersensitivity, bronchospasm, throat tightness, angioedema, edema (pharyngeal, palatal, mouth, peripheral, localized), swelling (face, eye, lip, tongue, upper airway), facial hypoesthesia, rash, urticaria, pruritus, flushing, and erythema have occurred in patients with no prior exposure to perflutrencontaining microsphere products.
- When administering OPTISON to patients with a cardiac shunt, microspheres can bypass filtering of the lungs and enter the arterial circulation. Assess patients with shunts for embolic phenomena following OPTISON administration. OPTISON is only for intravenous administration; do not administer OPTISON by intra-arterial injection.
- High ultrasound mechanical index values may cause microsphere rupture and lead to ventricular arrhythmias. Additionally, end-systolic triggering with high mechanical indices has been reported to cause ventricular arrhythmias. OPTISON is not recommended for use at mechanical indices greater than 0.8.
Adverse Reactions
Common adverse reactions (incidence > 0.5%) were: headache, nausea and/or vomiting, warm sensation or flushing, dizziness, dysgeusia, chills or fever, flu-like symptoms, malaise/weakness/fatigue, chest pain, dyspnea, injection site discomfort, and erythema. Cardiac arrests and other serious but nonfatal adverse reactions were uncommonly reported in postapproval use. Reports also identified neurologic reactions (loss of consciousness or convulsions) as well as anaphylactoid reactions. Overall, the safety profile observed in pediatric patients from the clinical study was consistent with the safety profile in adult patients.
Use in Specific Populations
Pregnancy and Lactation:
There are no data with OPTISON use in pregnant woman to inform any drug-associated risks. There are no data on the presence of perflutren protein-type A microspheres in human milk, the effects on the breastfed infant or the effects on milk production.
Pediatric Use
Safety and efficacy of OPTSON in pediatric patient is supported by evidence from adequate and well-controlled studies in adults and additional efficacy and safety data from a clinical study in 37 pediatric patients aged 9-17 years.
Geriatric Use
No overall differences in safety or effectiveness were observed in patients 65 years and over but a greater sensitivity to OPTISON in older individuals cannot be ruled out.
Please see the full Prescribing Information, including Boxed Warning for additional important safety information.
To report SUSPECTED ADVERSE REACTIONS, contact GE HealthCare at 800 654 0118 (option 2 then option 1) or by email at GPV.drugsafety@gehealthcare.com or FDA at 800 FDA 1088 or www.fda.gov/medwatch
Serious cardiopulmonary reactions, including fatalities, have occurred uncommonly during or following perflutren-containing microsphere administration. Most serious reactions occur within 30 minutes of administration
• Assess all patients for the presence of any condition that precludes OPTISON administration
• Always have resuscitation equipment and trained personnel readily available
OPTISON (Perflutren Protein-Type A Microspheres Injectable Suspension, USP)
INDICATIONS:
OPTISON is an ultrasound contrast agent indicated for use in adult and pediatric patients with suboptimal echocardiogram to opacify to the left ventricle to improve the delineation of the left ventricle endocardial borders.
CONTRAINDICATIONS:
OPTISON is contraindicated in patients with known or suspected hypersensitivity to perflutren or albumin.
WARNINGS AND PRECAUTIONS:
- Serious cardiopulmonary reactions including fatalities have occurred uncommonly during or shortly following perflutren-containing microsphere administration, typically within 30 minutes of administration. The risk for these reactions may be increased among patients with unstable cardiopulmonary conditions (acute myocardial infarction, acute coronary artery syndromes, worsening or unstable congestive heart failure, or serious ventricular arrhythmias).
- Serious anaphylactic reactions have been observed during or shortly following perflutrencontaining microsphere administration, including shock, hypersensitivity, bronchospasm, throat tightness, angioedema, edema (pharyngeal, palatal, mouth, peripheral, localized), swelling (face, eye, lip, tongue, upper airway), facial hypoesthesia, rash, urticaria, pruritus, flushing, and erythema have occurred in patients with no prior exposure to perflutrencontaining microsphere products.
- When administering OPTISON to patients with a cardiac shunt, microspheres can bypass filtering of the lungs and enter the arterial circulation. Assess patients with shunts for embolic phenomena following OPTISON administration. OPTISON is only for intravenous administration; do not administer OPTISON by intra-arterial injection.
- High ultrasound mechanical index values may cause microsphere rupture and lead to ventricular arrhythmias. Additionally, end-systolic triggering with high mechanical indices has been reported to cause ventricular arrhythmias. OPTISON is not recommended for use at mechanical indices greater than 0.8.
Adverse Reactions
Common adverse reactions (incidence > 0.5%) were: headache, nausea and/or vomiting, warm sensation or flushing, dizziness, dysgeusia, chills or fever, flu-like symptoms, malaise/weakness/fatigue, chest pain, dyspnea, injection site discomfort, and erythema. Cardiac arrests and other serious but nonfatal adverse reactions were uncommonly reported in postapproval use. Reports also identified neurologic reactions (loss of consciousness or convulsions) as well as anaphylactoid reactions. Overall, the safety profile observed in pediatric patients from the clinical study was consistent with the safety profile in adult patients.
Use in Specific Populations
Pregnancy and Lactation:
There are no data with OPTISON use in pregnant woman to inform any drug-associated risks. There are no data on the presence of perflutren protein-type A microspheres in human milk, the effects on the breastfed infant or the effects on milk production.
Pediatric Use
Safety and efficacy of OPTSON in pediatric patient is supported by evidence from adequate and well-controlled studies in adults and additional efficacy and safety data from a clinical study in 37 pediatric patients aged 9-17 years.
Geriatric Use
No overall differences in safety or effectiveness were observed in patients 65 years and over but a greater sensitivity to OPTISON in older individuals cannot be ruled out.
Please see the full Prescribing Information, including Boxed Warning for additional important safety information.
To report SUSPECTED ADVERSE REACTIONS, contact GE HealthCare at 800 654 0118 (option 2 then option 1) or by email at GPV.drugsafety@gehealthcare.com or FDA at 800 FDA 1088 or www.fda.gov/medwatch