Don't have an account?
At a glance
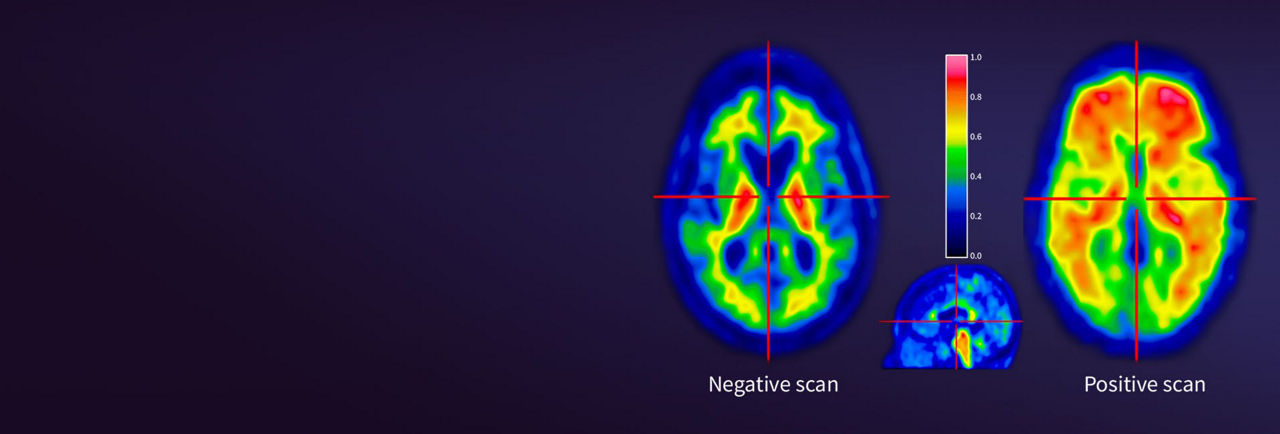
Comprehensive, clear results
Vizamyl is the only amyloid PET imaging agent with the following imaging and interpretation features that may help enhance diagnostic clarity¹
• Five defined read regions, including the striatum for assistance especially in cases of atrophy
• FDA approved to be read in full color scale (including more than 5 distinct colors)
• Single positive read region is sufficient to classify a scan positive
Consistent scan assessment
Unlike tracers that normalize to regions of highest uptake, which varies within and between patients, Vizamyl uses a standardized color scale anchored to the pons, a region minimally affected by amyloid. This provides a stable anchor point for consistent interpretation across patients and readers.¹⁻³
Precise, accurate quantification
As the only FDA-approved tracer derived from the in vivo imaging gold-standard Pittsburgh Compound B (PiB), Vizamyl supports a high dynamic range and delivers precise, accurate Centiloid (CL) quantification-enabling objective, reliable assessment of amyloid burden to inform diagnosis and patient management.1,4-7
Efficient workflows
Non-cerebellar uptake (i.e salivary gland uptake) can interfere with registration and quantification. Vizamyl's minimal non-cerebellar uptake reduces the need for manual reorientation supporting efficient workflows and accurate quantification results clinicians can trust.8-9
A clinical challenge
Up to
20% of scans
can be challenging or difficult to interpret due to a myriad of factors including atrophy or structural anomalies, borderline or emerging pathology, etc.4,8,10
With the advent of anti-amyloid therapies and the increasing demand for amyloid PET imaging, it’s more important than ever to assess amyloid burden clearly and consistently for confident diagnosis and treatment planning.
• Frontal lobes
• Posterior cingulate and precuneusv • Lateral temporal lobes
• Inferolateral parietal lobes
• Striatum (less affected by atrophy and can provide clarity especially for difficult scans)
Vizamyl also offers 3 imaging planes of view¹:
• Axial
• Sagittal
• Coronal
A single positive read region is sufficient to classify a Vizamyl scan as positive.¹
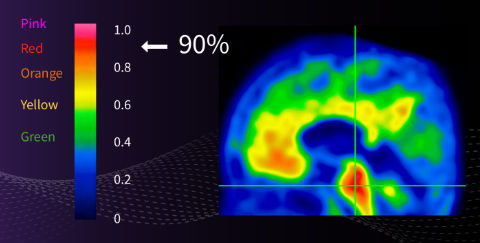
Pons set to 90% of maximum intensity¹
• Reference intensity for the rest of the brain
• Independent of cortex plaque load
Pittsburgh compound B (PiB), the in vivo gold standard for amyloid PET imaging was used to develop the Centiloid scale, a universal measure of amyloid burden.6,11
Vizamyl is the only amyloid PET imaging agent derived from PiB and supports a high dynamic range to deliver precise, accurate CL quantification for confident reads even in challenging cases.1,4-6
GE HealthCare provides the support you need to scan and interpret with confidence
References
- Vizamyl Prescribing Information. GE Healthcare; 2025.
- Eli Lilly and Company. Amyvid (Florbetapir F 18) Injection Prescribing Information. Published 2022. Accessed October 22, 2025.https://pi.lilly.com/us/amyvid-uspi.pdf.
- Life Molecular Imaging. Neuraceq (Fluorbetaben F18) Injection Prescribing Information. Published 2022. Accessed October 22, 2025. https://neuraceq.com/wp-content/uploads/PRESCRIBING- INFORMATION.pdf.
- Collij LE, Bischof GN, Altomare D, et al. Quantification supports amyloid PET visual assessment of challenging cases: results from the AMYPAD diagnostic and patient management study. J Nucl Med. 2024. doi:10.2967/jnumed.124.268119. [jnm.snmjournals.org]
- Pemberton HG, Collij LE, Heeman F, et al. Quantification of amyloid PET for future clinical use: a state-of-the-art review. Eur J Nucl Med Mol Imaging. 2022;49(10):3508-28.
- Myburgh PJ, Solingapuram Sai KK. Two decades of [11C]PiB synthesis, 2003–2023: a review. Am J Nucl Med Mol Imaging. 2024;14(1):48–62. Available from: https://e-century.us/files/ajnmmi/14/1/ajnmmi0154938.pdf.
- Rabinovici GD, et al. Updated appropriate use criteria for amyloid and tau PET. Alzheimer’s Dement. 2025;21:e14338. doi:10.1002/alz.14338.
- Schmidt ME, Chiao P, Klein G, et al. The influence of biological and technical factors on quantitative analysis of amyloid PET: points to consider and recommendations for controlling variability in longitudinal data. Alzheimers Dement. 2015;11(9):1050–1068. doi:10.1016/j.jalz.2014.09.004.
- European Medicines Agency. Amyvid: EPAR – Product Information. EMA. Published October 2021. Accessed October 22, 2025. https://www.ema.europa.eu/en/documents/product-information/ amyvid-epar-productinformation_ en.pdf.
- Jagust WJ, Mattay VS, Krainak DM, et al. Quantitative brain amyloid PET. J Nucl Med. 2024;65(5):670–678. doi:10.2967/jnumed.123.265766.
- Klunk WE, Koeppe RA, Price JC, et al. The Centiloid project: standardizing quantitative amyloid plaque estimation by PET. Alzheimers Dement. 2015;11(1):1–15. doi: 10.1016/j.jalz.2014.07.003.
PRODUCT INDICATIONS AND USE
VIZAMYL™ (flutemetamol F 18 injection) is indicated for positron-emission tomography (PET) of the brain to estimate amyloid beta neuritic plaque density in adults with cognitive impairment for:
- Evaluation of Alzheimer’s disease (AD) and other causes of cognitive decline
- Selection of patients who are indicated for amyloid beta-directed therapy as described in the prescribing of information of the therapeutic products.
CONTRAINDICATIONS
- VIZAMYL is contraindicated in patients with a history of hypersensitivity reaction to VIZAMYL or polysorbate 80.
WARNINGS AND PRECAUTIONS
- Anaphylaxis and Other Serious Hypersensitivity Reactions: Serious hypersensitivity reactions including anaphylaxis, presenting with flushing, dyspnea, and hypotension have been observed within minutes following administration and may occur in patients with no history of exposure to VIZAMYL. Obtain a history of allergy or hypersensitivity reactions. Always have resuscitation equipment and trained personnel immediately available at the time of VIZAMYL administration. If a hypersensitivity reaction is suspected, immediately discontinue the injection and initiate appropriate therapy. VIZAMYL is contraindicated in patients with a history of hypersensitivity to VIZAMYL or polysorbate 80.
- Risk of Image Misinterpretation and Other Errors: Errors may occur in the estimation of amyloid beta neuritic plaque density during VIZAMYL image interpretation. The use of clinical information in the interpretation of VIZAMYL images has not been evaluation and may lead to an inaccurate assessment. Extensive brain atrophy and motion artifacts that distort the image may limit the ability to distinguish gray and white matter on a VIZAMYL scan. Perform image interpretation independently of the patient’s clinical information. For cases where there is uncertainty as to the location of cortical signal, use co-registered anatomical imaging to improve localization of signal or examine the striatum for VIZAMYL signal as it is less affected by atrophy.
- Radiation Risk: VIZAMYL contributes to a patient’s long-term cumulative radiation exposure. Long-term cumulative radiation exposure is associated with an increased risk of cancer. Ensure safe drug handling to protect patients and health care providers from unintentional radiation exposure. Advise patients to hydrate before and after administration and to void frequently after administration.
ADVERSE REACTIONS
- The most commonly reported adverse reactions in clinical trials were flushing (2 %), increased blood pressure (2 %), headache (1 %), nausea and dizziness (1 %). Postmarketing experience included anaphylactic reactions. Because these reactions are reported voluntarily from a population of uncertain size, it is not always possible to reliably estimate their frequency or establish a causal relationship to drug exposure.
USE IN SPECIFIC POPULATIONS
- Pregnancy: There are no available data on VIZAMYL in pregnant woman to evaluate drug associated risk of major birth defects, miscarriage or adverse maternal or fetal outcomes. All radiopharmaceuticals, including VIZAMYL, have the potential to cause fetal harm depending on the stage of fetal development and the magnitude of the radiation dose. If considering VIZAMYL administration to a pregnant woman, inform the patient about the potential for adverse pregnancy outcomes based on the radiation dose from the drug and the gestational timing of exposure.
- Lactation: There are no data on the presence of flutemetamol F 18 or metabolites in human milk or its effects on the breastfed infant or milk production. Exposure of VIZAMYL to a breastfed infant can be minimized by temporary discontinuation of breastfeeding. The developmental and health benefits of breastfeeding should be considered along with the mother’s clinical need for VIZAMYL and any potential adverse effects on the breastfed child from VIZAMYL or from the underlying maternal condition.
- Pediatric Use: The safety and effectiveness of VIZAMYL have not been established pediatric patients.
- Geriatric Use: No overall differences in safety or effectiveness were observed between subjects 65 years of age and older and younger adult subjects.
OVERDOSAGE
- The major risks of overdosage relate predominantly to increased radiation exposure, with long-term risk for neoplasia. In the event of administration of a radiation overdose with VIZAMYL, hydration and frequent urination should be encouraged to minimize radiation exposure to the subject. It is unknown whether or not the flutemetamol is dialyzable.
Prior to VIZAMYL administration, please read the full Prescribing Information for additional Important Safety Information.
To report SUSPECTED ADVERSE REACTIONS, contact GE HealthCare at 800 654 0118 (option 2 then option 1) or by email at GPV.drugsafety@gehealthcare.com or FDA at 800 FDA 1088 or www.fda.gov/medwatch
PRODUCT INDICATIONS AND USE
VIZAMYL™ (flutemetamol F 18 injection) is indicated for positron-emission tomography (PET) of the brain to estimate amyloid beta neuritic plaque density in adults with cognitive impairment for:
- Evaluation of Alzheimer’s disease (AD) and other causes of cognitive decline
- Selection of patients who are indicated for amyloid beta-directed therapy as described in the prescribing of information of the therapeutic products.
CONTRAINDICATIONS
- VIZAMYL is contraindicated in patients with a history of hypersensitivity reaction to VIZAMYL or polysorbate 80.
WARNINGS AND PRECAUTIONS
- Anaphylaxis and Other Serious Hypersensitivity Reactions: Serious hypersensitivity reactions including anaphylaxis, presenting with flushing, dyspnea, and hypotension have been observed within minutes following administration and may occur in patients with no history of exposure to VIZAMYL. Obtain a history of allergy or hypersensitivity reactions. Always have resuscitation equipment and trained personnel immediately available at the time of VIZAMYL administration. If a hypersensitivity reaction is suspected, immediately discontinue the injection and initiate appropriate therapy. VIZAMYL is contraindicated in patients with a history of hypersensitivity to VIZAMYL or polysorbate 80.
- Risk of Image Misinterpretation and Other Errors: Errors may occur in the estimation of amyloid beta neuritic plaque density during VIZAMYL image interpretation. The use of clinical information in the interpretation of VIZAMYL images has not been evaluation and may lead to an inaccurate assessment. Extensive brain atrophy and motion artifacts that distort the image may limit the ability to distinguish gray and white matter on a VIZAMYL scan. Perform image interpretation independently of the patient’s clinical information. For cases where there is uncertainty as to the location of cortical signal, use co-registered anatomical imaging to improve localization of signal or examine the striatum for VIZAMYL signal as it is less affected by atrophy.
- Radiation Risk: VIZAMYL contributes to a patient’s long-term cumulative radiation exposure. Long-term cumulative radiation exposure is associated with an increased risk of cancer. Ensure safe drug handling to protect patients and health care providers from unintentional radiation exposure. Advise patients to hydrate before and after administration and to void frequently after administration.
ADVERSE REACTIONS
- The most commonly reported adverse reactions in clinical trials were flushing (2 %), increased blood pressure (2 %), headache (1 %), nausea and dizziness (1 %). Postmarketing experience included anaphylactic reactions. Because these reactions are reported voluntarily from a population of uncertain size, it is not always possible to reliably estimate their frequency or establish a causal relationship to drug exposure.
USE IN SPECIFIC POPULATIONS
- Pregnancy: There are no available data on VIZAMYL in pregnant woman to evaluate drug associated risk of major birth defects, miscarriage or adverse maternal or fetal outcomes. All radiopharmaceuticals, including VIZAMYL, have the potential to cause fetal harm depending on the stage of fetal development and the magnitude of the radiation dose. If considering VIZAMYL administration to a pregnant woman, inform the patient about the potential for adverse pregnancy outcomes based on the radiation dose from the drug and the gestational timing of exposure.
- Lactation: There are no data on the presence of flutemetamol F 18 or metabolites in human milk or its effects on the breastfed infant or milk production. Exposure of VIZAMYL to a breastfed infant can be minimized by temporary discontinuation of breastfeeding. The developmental and health benefits of breastfeeding should be considered along with the mother’s clinical need for VIZAMYL and any potential adverse effects on the breastfed child from VIZAMYL or from the underlying maternal condition.
- Pediatric Use: The safety and effectiveness of VIZAMYL have not been established pediatric patients.
- Geriatric Use: No overall differences in safety or effectiveness were observed between subjects 65 years of age and older and younger adult subjects.
OVERDOSAGE
- The major risks of overdosage relate predominantly to increased radiation exposure, with long-term risk for neoplasia. In the event of administration of a radiation overdose with VIZAMYL, hydration and frequent urination should be encouraged to minimize radiation exposure to the subject. It is unknown whether or not the flutemetamol is dialyzable.
Prior to VIZAMYL administration, please read the full Prescribing Information for additional Important Safety Information.
To report SUSPECTED ADVERSE REACTIONS, contact GE HealthCare at 800 654 0118 (option 2 then option 1) or by email at GPV.drugsafety@gehealthcare.com or FDA at 800 FDA 1088 or www.fda.gov/medwatch