Don't have an account?
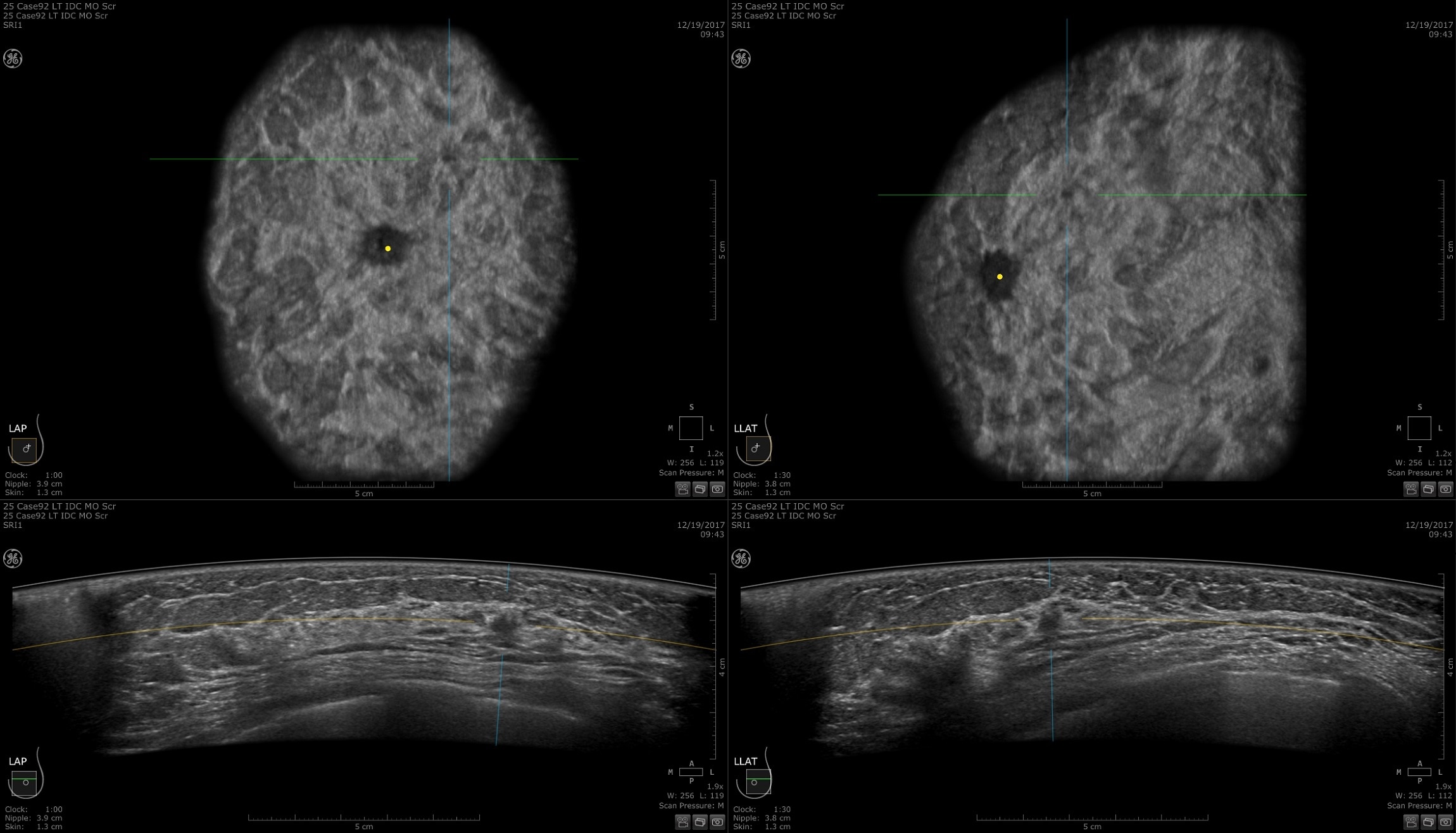
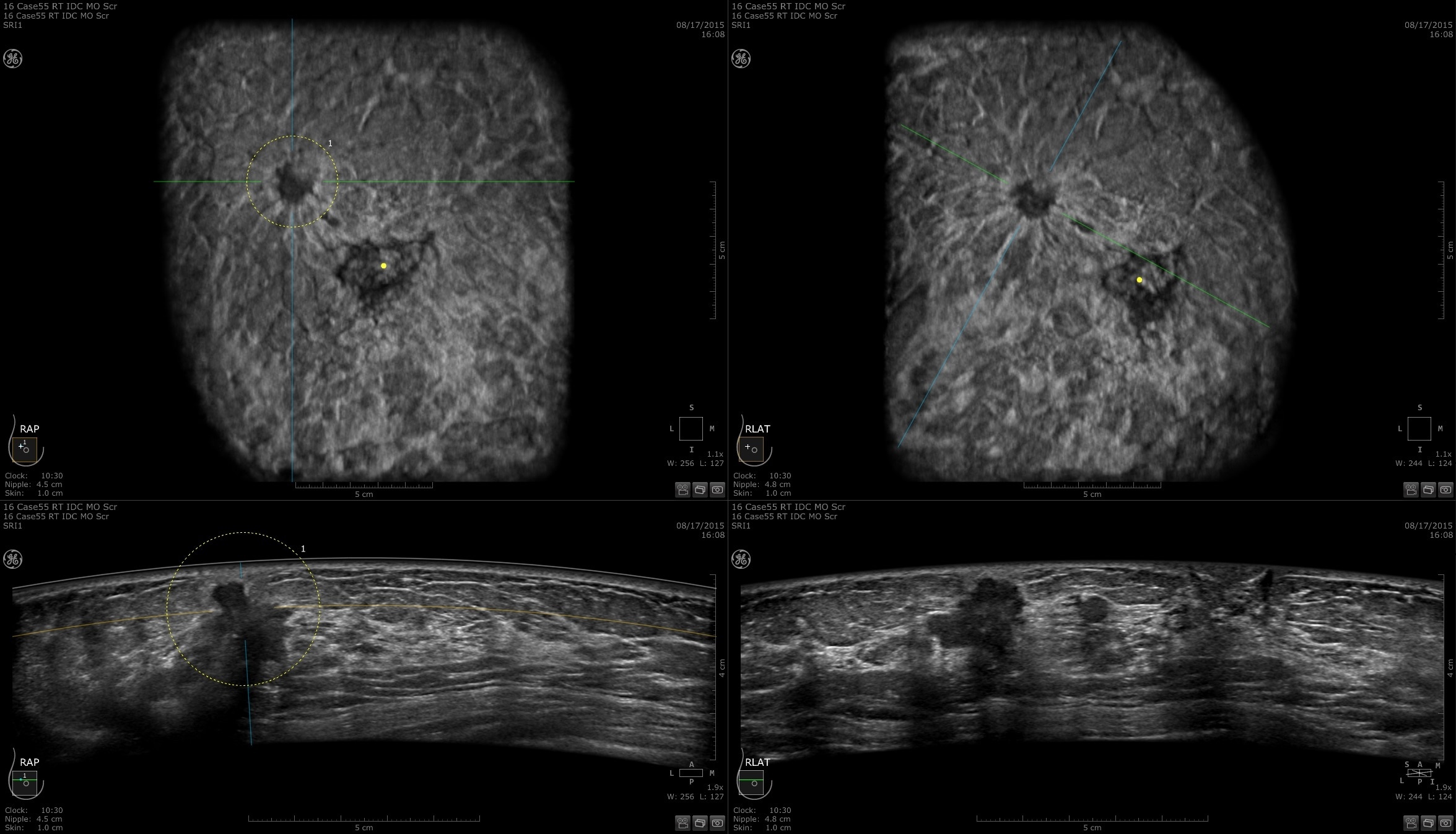
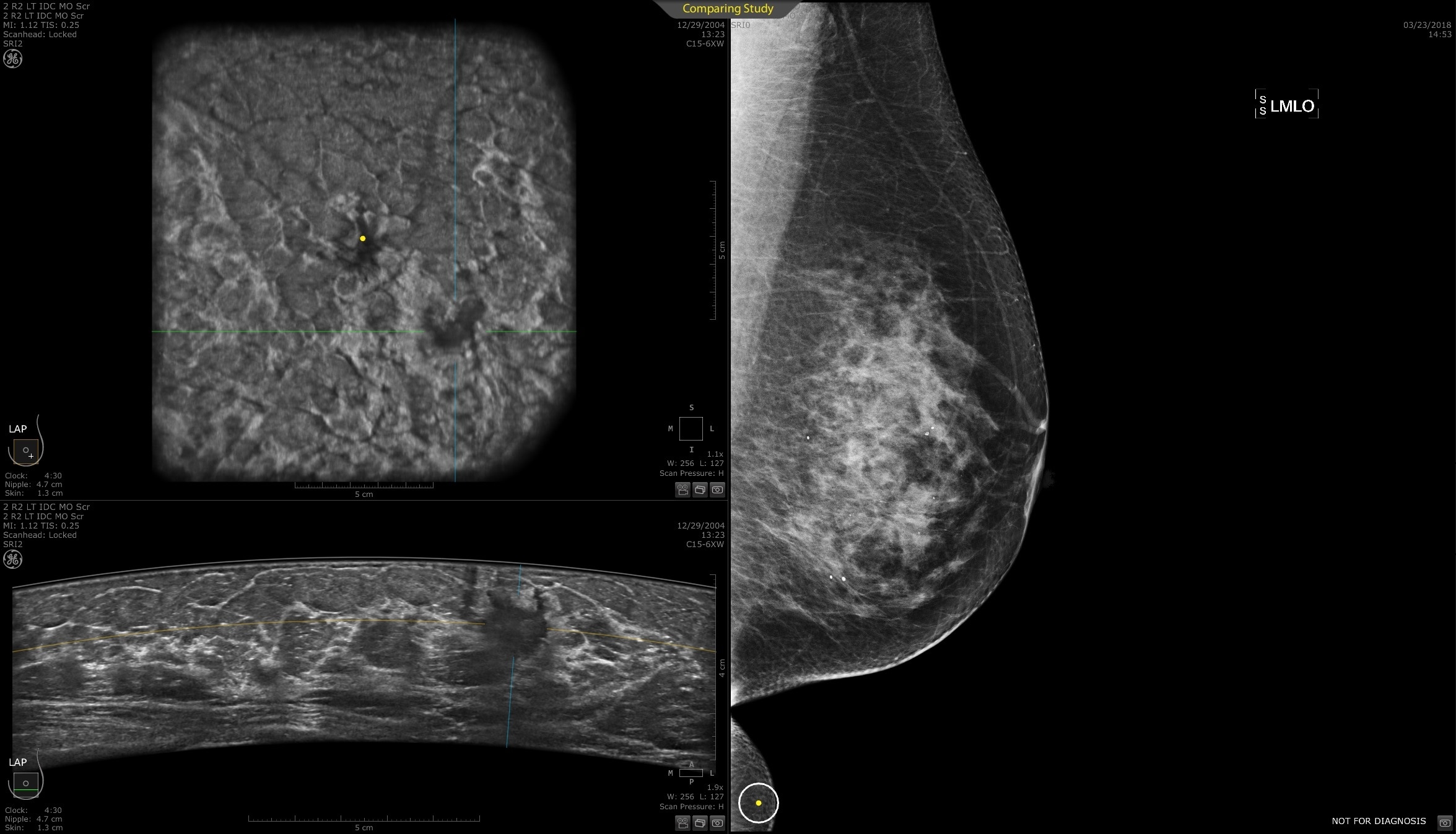
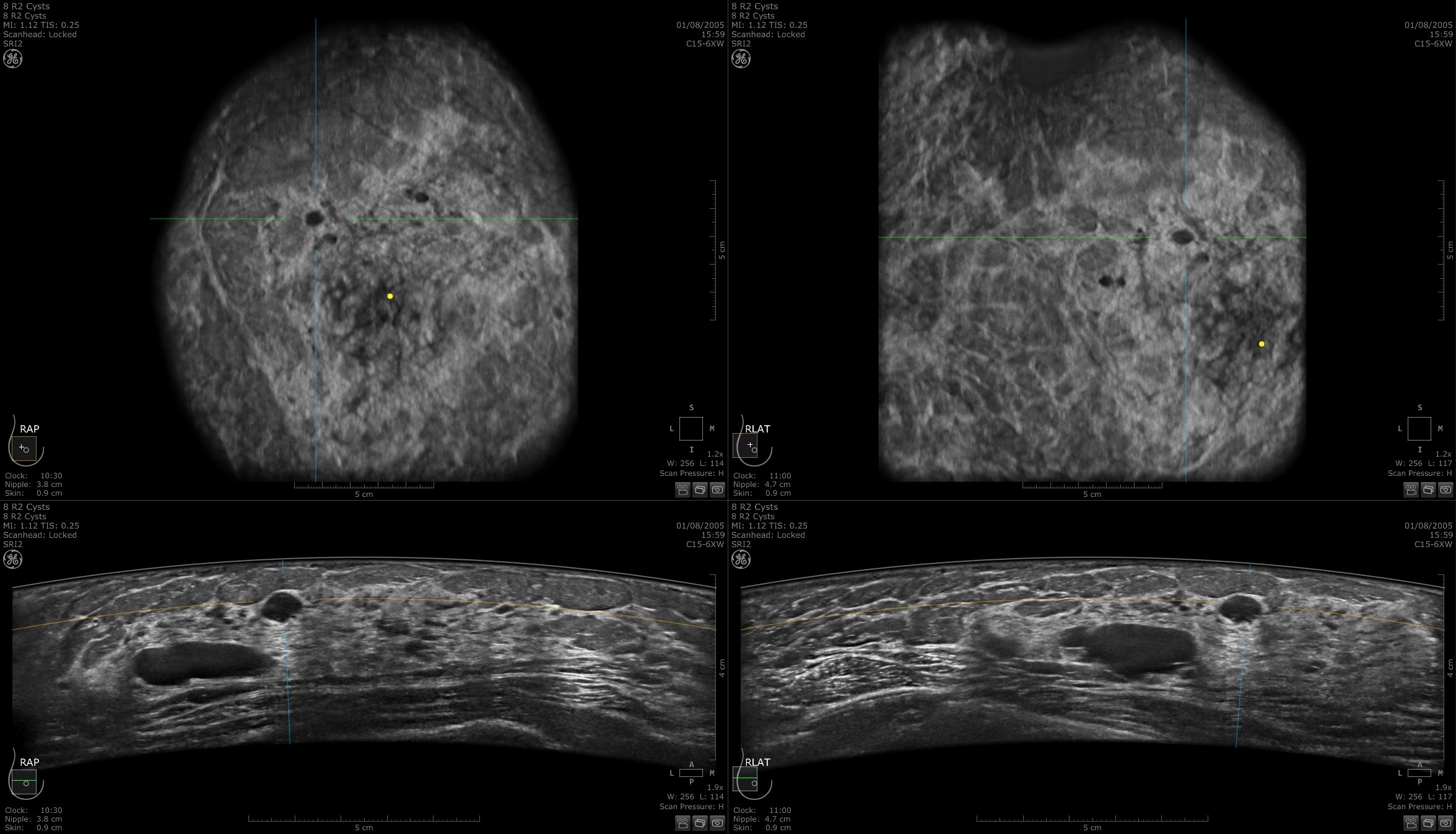
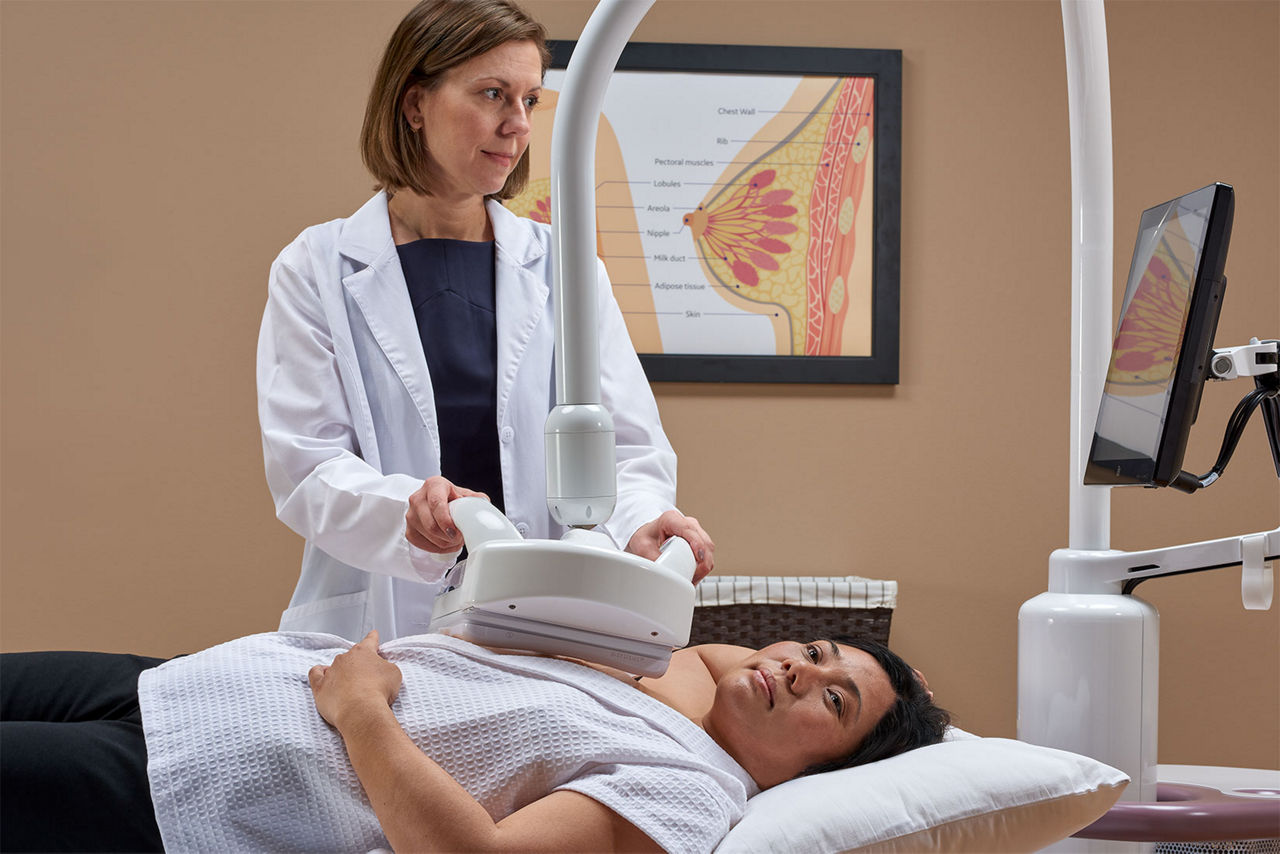
Designed for screening
Improve breast cancer detection by 35.7 percent over mammography alone1
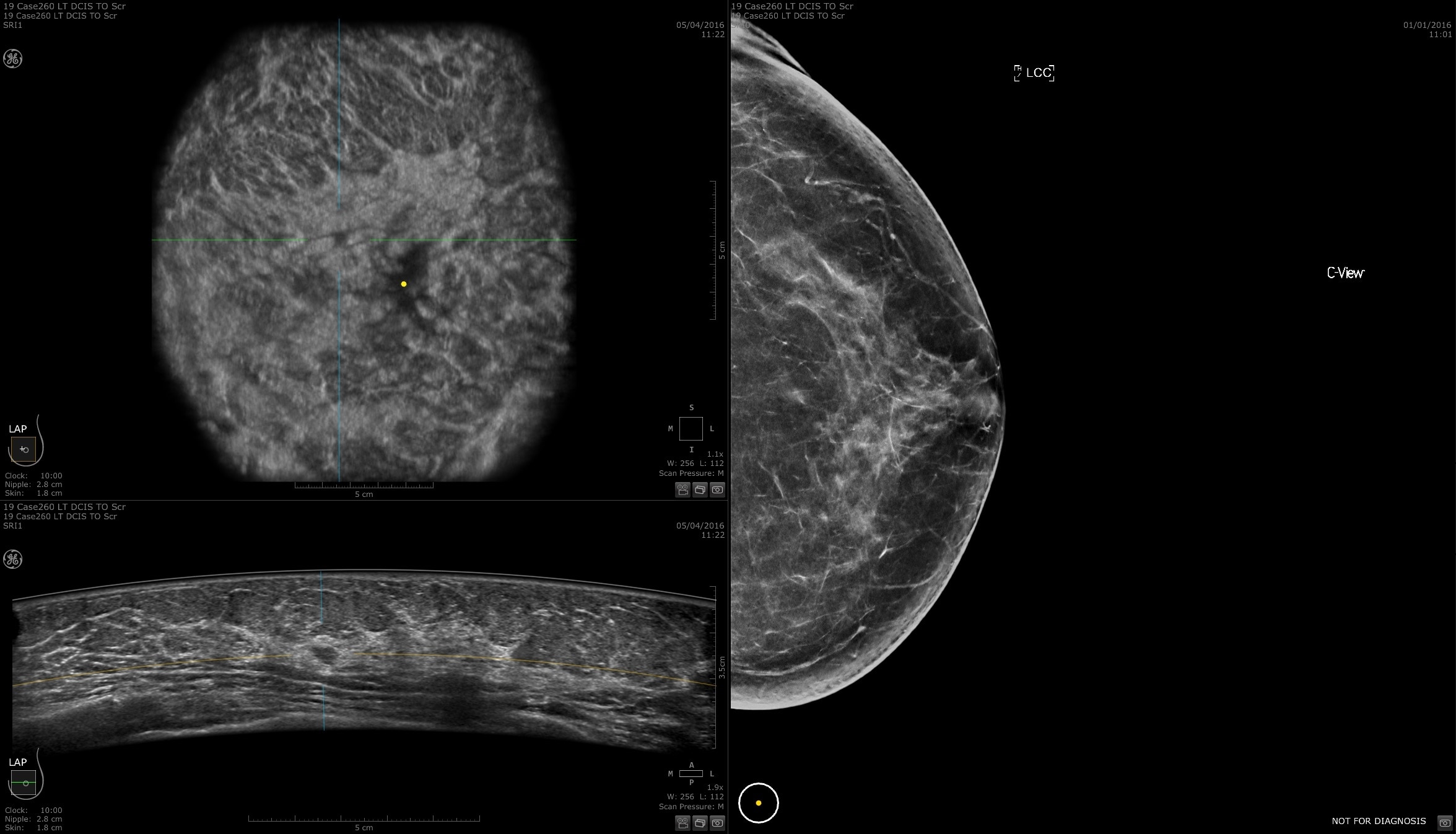
Remarkable image quality
Look differently at dense breast tissue with advanced interpretation tools
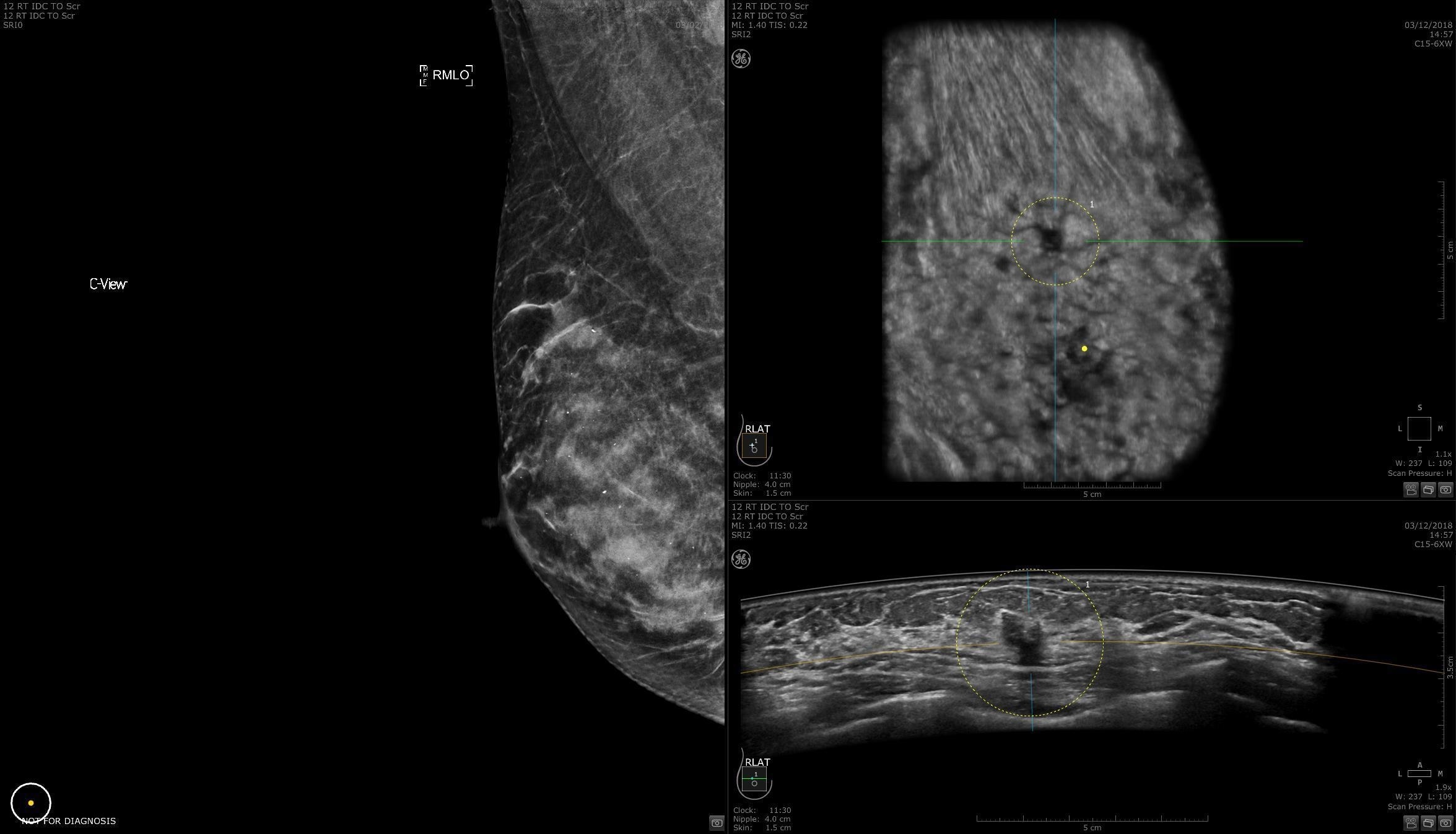
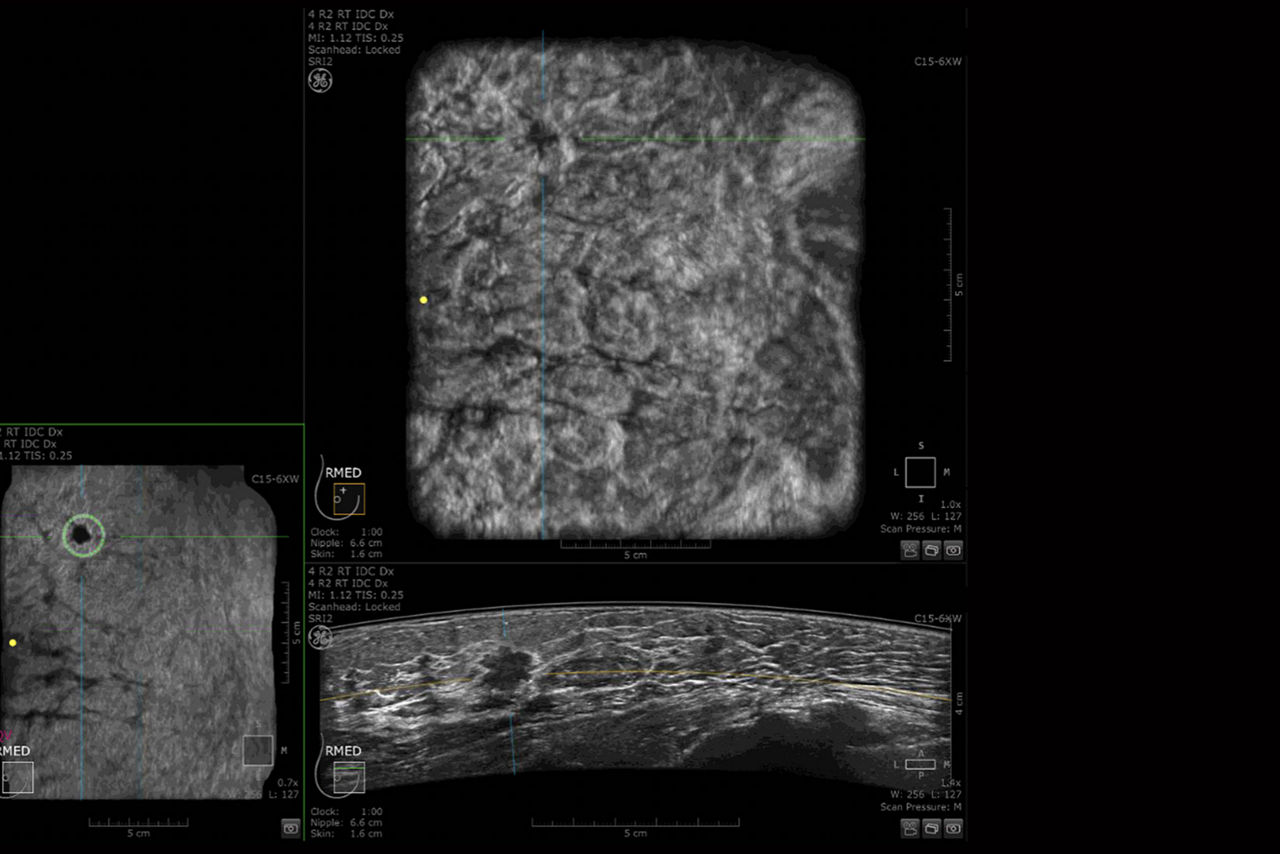
AI Assistant
Helps to detect and characterize breast lesions for clinical confidence