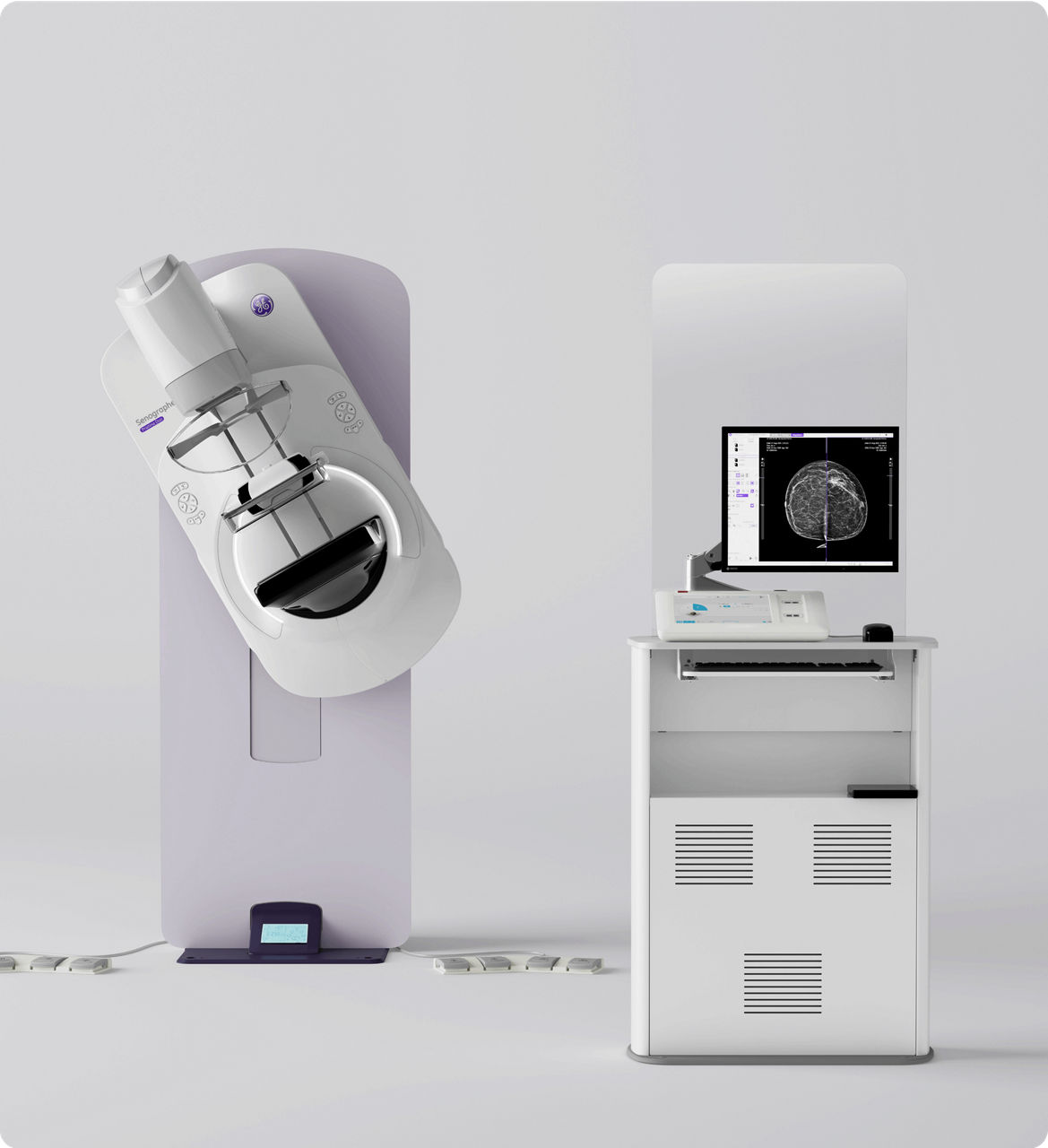
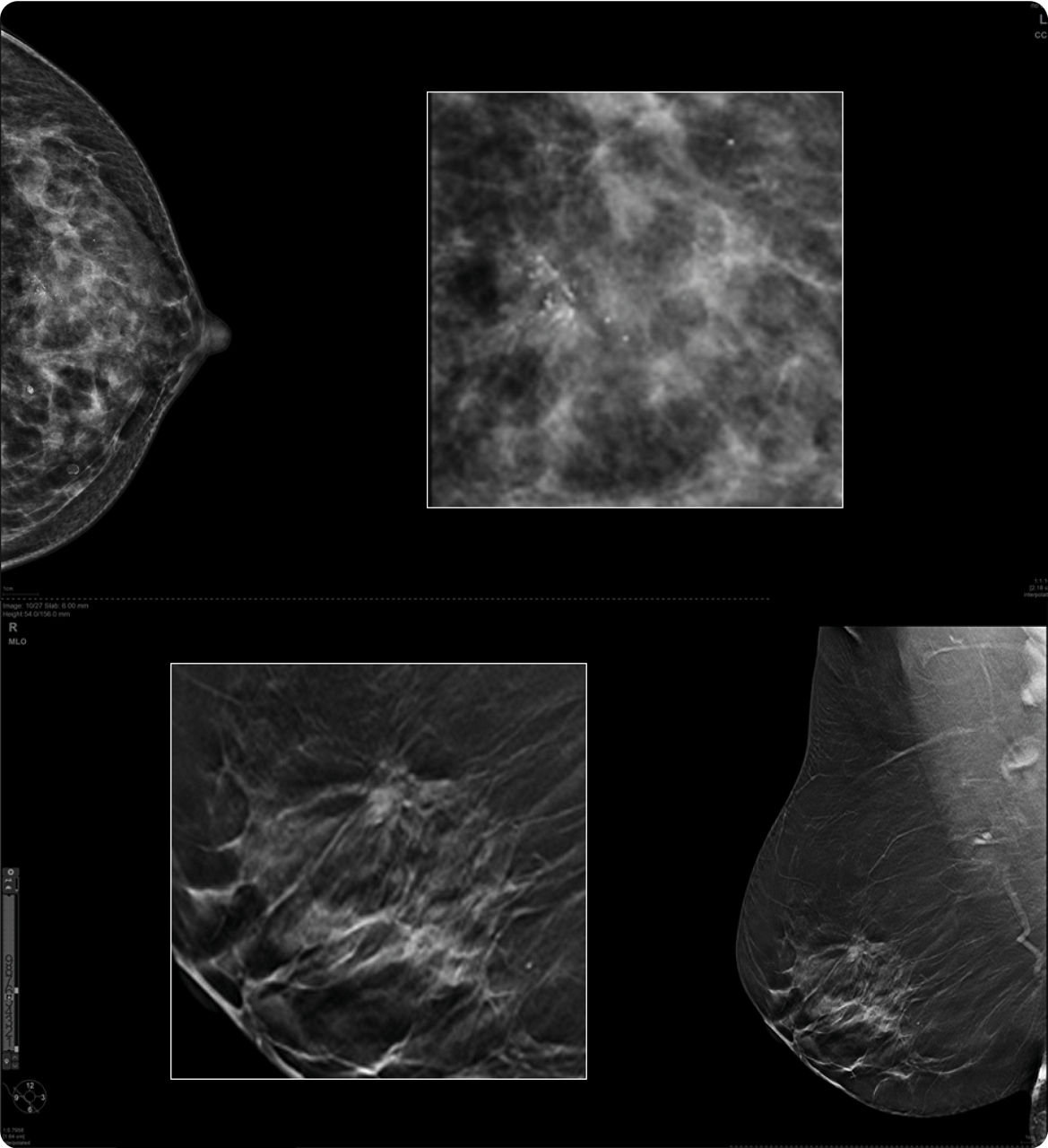
Pristina Duo is a commercial offering of Senographe Pristina
TM. All features and options may not be available in all countries. Please check with your local GE HealthCare representative for availability.
1. GE HealthCare DBT delivers superior diagnostic accuracy demonstrated in a reader study comparing
the ROC AUC of GE HealthCare screening protocol (V-Preview + 3D CC/MLO with 3D in STD mode) to that of 2D FFDM
alone. FDA PMA P130020.
2. IPSOS Technologists Study sponsored by GE HealthCare, conducted with 50 users in Japan and the
European Union, from July 2017 to April 2018.
3. IPSOS Patient Satisfaction Study sponsored by GE HealthCare, conducted with 315 patients across
2 sites in Europe, February 2017.
4. Analysis of 2,654 systems covering >2 million exams, Pristina Duo supports customers with the potential
to image up to 150 patients a day. Results may vary according to hospital size, working days, working hours, etc. there can be no guarantee that each Pristina system will achieve the same results.
5. Analysis of the 42,543 Service requests on Pristina platform systems under Contract or Warranty from January 2022 to September 2024. Results may vary according to hospital size, working days, working hours, etc . there can be no guarantee that each Pristina system will achieve the same results.
6. IPSOS Patient Satisfaction Study sponsored by GE HealthCare, conducted with 215 patients across
1 site in Italy, February 2017.
7. Phantom measurements as of July 2024. Senographe Pristina with default value STD mode. Dose comparison based on NHS Breast Screening Programme equipment reports.
https://medphys.royalsurrey.nhs.uk/nccpm/?s=technicalreports. Systems comparison in the USA.
8. At equivalent sensitivity. Superior diagnostic accuracy demonstrated in a reader study comparing the ROC AUC of GE HealthCare screening
protocol (V-Preview + 3D CC/MLO with 3D in STD mode) to that of 2D FFDM alone. V-Preview is the 2D synthesized image generated by GE HealthCare Seno Iris mammography software from GE HealthCare DBT images. FDA PMA P130020
http://www.accessdata.fda.gov/scripts/cdrh/cfdocs/cfpma/pma.cfm?id=P130020.
9. Digital Mammography versus Digital Mammography plus tomosynthesis for Breast Cancer Screening: The Reggio Emilia Tomosynthesis Randomized Trial.
https://pubs.rsna.org/doi/10.1148/radiol.2018172119. Applicable for Europe only.